Show that (11.6.4) follows from (11.6.3), (11.6.2), and the fermion anticommutation relations. |pair) = Akk bab|z|0), 10
Question:
Show that (11.6.4) follows from (11.6.3), (11.6.2), and the fermion anticommutation relations.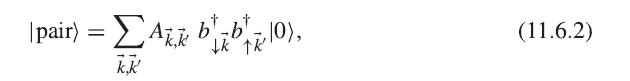
![]()
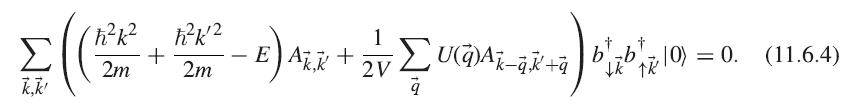
Transcribed Image Text:
|pair) = ΣAkk bab|z|0), 10 k,k' (11.6.2)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
H The Hamiltonian is bt b k 2m tk tk k and the pair state is k k bt 2m ktk b 2V U b 4 b b kk...View the full answer

Answered By

Sidharth Jain
My name is Sidharth. I completed engineering from National Institute of Technology Durgapur which is one of the top college in India. I am currently working as an Maths Faculty in one of the biggest IITJEE institute in India. Due to my passion in teaching and Maths, I came to this field. I've been teaching for almost 3 years.
Apart from it I also worked as an Expert Answerer on Chegg.com. I have many clients from USA to whom I teach online and help them in their assignments. I worked on many online classes on mymathlab and webassign. I guarantee for grade 'A'.
4.90+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
The data show the number of years of experience the players on the Pittsburgh Steelers football team have at the beginning of the season. Draw and analyze a dot plot for the data. 3 12 6. 12 5 1 4 5...
-
A certain university has decided to introduce the use of plus and minus with letter grades, as long as there is evidence that more than 60% of the faculty favor the change. A random sample of faculty...
-
Huddell Company, which is both a wholesaler and retailer, purchases merchandise from various suppliers. The dollar-value LIFO method is used for the wholesale inventories. Huddell determines the...
-
A milk company has four machines that fill gallon jugs with milk. The quality control manager is interested in determining whether the average fill for these machines is the same. The following data...
-
Suppose you raise this book (inertia \(m=3.4 \mathrm{~kg}\) ) from the floor to your desk, \(1.0 \mathrm{~m}\) above the floor. (a) Does the gravitational potential energy of the Earth-book system...
-
Coca-Cola and PepsiCo are the leading competitors in the market for cola products. In 1960 Coca-Cola introduced Sprite, which today is the worldwide leader in the lemon-lime soft drink market and...
-
23. A convex mirror used for rear view on an automobile has a radius of curvature of 3m. If a bus is located at 5m from this mirror, find the position, nature and magnification of the image. 24. An...
-
Plot k 2 (A 2 k )/(1 A 2 k ) vs k assuming constant interaction vertex U, for various choices of U, to see what the distribution of excited particles looks like at T = 0.
-
Determine the critical number of particles for condensation at a fixed temperature in a three-dimensional harmonic potential with energy U = 1/2kr 2 . The quantum states in a harmonic potential are...
-
Equipment was purchased on January 2, 2025, for $24,000, but no portion of the cost has been charged to depreciation. The corporation wishes to use the straight-line method for these assets, which...
-
Explain briefly how and what unemployment and inflation measure. What potential economic issues result from high levels of unemployment and inflation? Are there ideal 'target' rates for both of these...
-
The differences between U.S. GAAP and IFRS with respect to property, plant, and equipment, which do you believe is going to be the greatest challenge to converge? Why? What do you believe is the best...
-
JP Morgan has a debt-equity ratio of .45. The firm is assessing a new project that requires an initial cash outlay of $250,000 for equipment. The flotation cost is 9.1 percent for equity and 4.4...
-
Gel Shell makes gel nail polish, which they sell for $18/bottle. Gel Shell has the following costs Gel Shell's Costs Type of Cost Amount Direct Materials $2/unit Direct Labor $3/unit Variable...
-
Carolina Lab Supply sells a dual access laminar flow hood with a list price of $60,000 to BioStart Inc. BioStart Inc. will pay $65,000 in one year and gives Carolina a note payable. This type of note...
-
The structure of tartaric acid is a. Is the form of tartaric acid pictured below optically active? Explain. b. Draw rhe optically active forms of tartaric acid. OH OH HO2C-CH CH-CO,H OH OH C-C
-
Draw the major product for each of the following reactions: (a) (b) (c) 1) 9-BBN 2) H2O2, NaOH 1) Disiamylborane 2) H20, NaOH
-
For each of the following IR spectra, determine whether it is consistent with the structure of a ketone, an alcohol, a carboxylic acid, a primary amine, or a secondary amine. a. b. c. d. e. f. 100-...
-
Carefully consider the structure of 2, 3-dimethyl-2-butene. There are twelve C sp3 H bonds, and they are all identical. Nevertheless, there is more than one signal just to the right of 3000 cm -1 in...
-
Match each compound with the appropriate IR spectrum: a. b. c. d. e. f. `NH2 100- 60- 20- 4000 3500 3000 2500 2000 1500 1000 Wavenumber (cm-1) % Transmittance
-
Write a program that first asks user to enter a lower limit integer and an upper limit integer, respectively. Then print out all integers between the user-entered lower limit and upper limit...
-
6. Consider the linear system 3x1 +52 = 3 3x1 + 4x2 = 4 2x1 +32=5 (a) Find the projection b, of the vector of the right hand sides to the column space of the coefficient matrix. (b) Compute the...
-
5 ) Beginning in year 0 , with a payment of $ 5 0 , 0 0 0 , what is the present value of an asset in perpetuity that appreciates by 4 % each year and which is discounted at the rate of 6 % each year?...

Study smarter with the SolutionInn App


