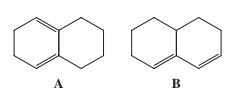
Bicyclic diene A reacts readily with appropriate alkenes by the Diels-Alder reaction, whereas diene B is totally
Question:
Bicyclic diene A reacts readily with appropriate alkenes by the Diels-Alder reaction, whereas diene B is totally unreactive. Explain.
Transcribed Image Text:
A B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
Since in A the conjugated double bonds are in the same side ie ...View the full answer

Answered By

Rama Rathi
With more than several years of teaching experience, I will take care of the concept building of students.
After being stood as a 1st rank holder in my school, I can also help you in a way that you can easily get good marks.
In my works I will try to ellaborate the concepts and make you strong enough to face similar problems easily.
Besides this I will also thake care of the customer satisfaction at a very affordable price.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Show the product of the Diels?Alder reaction of the following diene with 3-buten-2-one, H 2 C = CHCOCH 3 . Make sure you show the full stereochemistry of the reaction product.
-
Acetylenic compounds may be used as dienophiles in the Diels-Alder reaction (see Practice Problem 13.16). Write structures for the adducts that you expect from the reaction of 1,3-butadiene with (a)...
-
When 1, 3-cyclopentadiene and maleic anhydride (Eq. 15.12a, p.698) are allowed to react at room temperature, a Diels-Alder reaction takes place in which the endo product is formed as the major...
-
Another liability that employers have is that of unemployment insurance. Class, let's talk about this a little more. At what levels are unemployment liabilities incurred? What are some additional...
-
List and explain the four perspectives the balanced scorecard uses to analyze an organization's performance. How does this relate to project selection?
-
Consider the following data concerning the Poussin company: States Probabilits Market returns Profitability of the title 1 0,1 -0,18 -0,32 2 0,3 0,07 0,00 3 0,4 0,16 0,22 4 0,2 0,21 0,40 The...
-
Which backup method is the fastest? a. full daily backup c. differential daily backup b. incremental daily backup d. complete daily backup
-
The FASB has defined several terms in regard to accounting for income taxes. Below are various code letters (for terms) followed by definitions. ______1. The deferred tax consequences of future...
-
Your textdiscusses "termination" of the counseling relationship. Some contemporary counselors are taking a unique position with regard to termination. It goes something like this: If you have a...
-
Table 1 shows Apple's online orders for the last week. When shoppers place an online order, several "recommended products" (upsells) are shown as at checkout an attempt to upsell See table 2 in cell...
-
Dimethyl azodicarboxylate (see margin) takes part in the Diels-Alder reaction as a dienophile. Write the structure of the product of cycloaddition of this molecule with each of the following dienes....
-
Formulate the expected product of each of the following reactions. H;CO. H,C CH3 hv hv () (b) () (d) D CH3 H H;C H. CH;
-
Do male and female communication patterns differ regarding verbal dominance?
-
What is the PCAOB and what is its mandate?
-
A bank is trading on its own account \($10m\) of corporate bonds and \($5m\) of Treasuries. The daily volatility of corporate bonds is 1 =0.9%, and the daily volatility of Treasuries is 2 = 0.6%....
-
The COSO organization has issued a document describing a com- prehensive approach to Enterprise Risk Management (ERM). Required a. What are the major elements contained in the definition of ERM? b....
-
An auditor suspects that fictitious sales may have been recorded during the year. Which of the following analytical review results would most likely indicate that fictitious sales were recorded? a....
-
Comparing client data with industry data and with its own results for the previous year, the auditor finds that the number of days' sales in accounts receivable for this year is 66 for the client, 42...
-
Calculate the number of atoms per cubic meter in Pb?
-
At 31 December 20X9, the end of the annual reporting period, the accounts of Huron Company showed the following: a. Sales revenue for 20X9, $ 2,950,000, of which one- quarter was on credit. b....
-
Describe the physical significance of the partition function.
-
Enumerate the ways by which the parameter f3 may be identified with 1/kT.
-
Explain what is meant by an ensemble and why it is useful in statistical thermodynamics.
-
A southern California company manufactures surfboards. The company uses a job-order costing system with a single plantwide predetermined overhead rate based on machine hours. The predetermined...
-
Discuss the factors that play a vital role in Capacity Planning and Expansion at a Distribution Center/Warehouse
-
By improving its net patient receivables management, Jim Medical Center increased its cash position by $3,420,000 between fiscal year 2018 and fiscal year 2019. Net patient revenues in 2018 and 2019...

Study smarter with the SolutionInn App


