Formulate the product(s) that you would expect from each of the following reactions. Show stereochemistry clearly. Ch
Question:
Formulate the product(s) that you would expect from each of the following reactions. Show stereochemistry clearly.
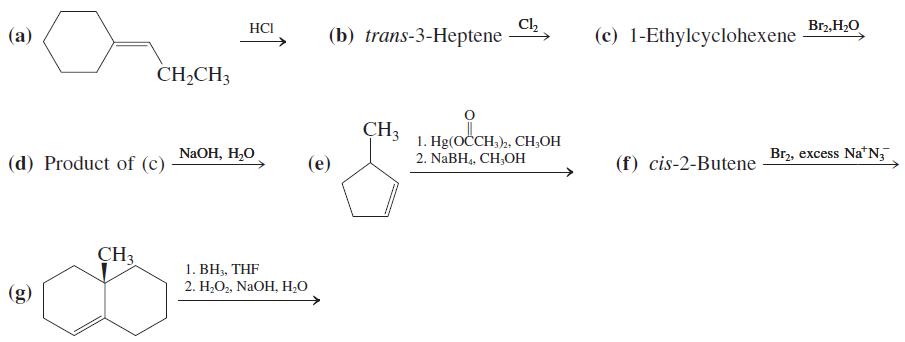
Transcribed Image Text:
Ch (b) trans-3-Heptene HCI Br2, H2O (a) (c) 1-Ethylcyclohexene CH,CH3 CH3 NaOH, H,O 1. Hg(OČCH,)2, CH,OH 2. NABH,, CH,OH Br2, excess Na'N, (d) Product of (c) (e) (f) cis-2-Butene CH3 1. BH3, THF 2. H;O,, NaOH, H2O (g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (12 reviews)
the expla...View the full answer

Answered By

YOGENDRA NAILWAL
As I'm a Ph.D. student, so I'm more focussed on my chemistry laboratory. I have qualified two national level exams viz, GATE, and NET JRF (Rank 68). So I'm highly qualified in chemistry subject. Also, I have two years of teaching experience in this subject, which includes college teacher as well as a personal tutor. I can assure you if you hire me on this particular subject, you are never going to regret it.
Best Regards.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
What products would you expect from each of the following reactions? (a) (b) (c) t-BuOK CHBr CH2Zn(Cu) diethyl ether
-
Write stereochemical formulas for all of the products that you would expect from each of the following reactions. (You may find models helpful.) (a) (b) (c) (d) (1) OsO4 (2) NaHSO3, H2O (1) OsO (2)...
-
Formulate the product of each of the following reactions. The solvent in each case is (CH3CH2)2O. (a) (b) (c) O .LiAID 2. H, H,O CH3CH O 1.LIAIH 2D D.O LiAID CH,CH>
-
A- Farah Corporation has provided the following production and total cost data for two levels of monthly production volume. The company produces a single product which it sells at $132.7 per unit...
-
Describe the drawbacks to using qualitative risk analysis techniques.
-
How does the United States Commercial Service help small businesses abroad specifically?
-
Return on Sales Calculate the company's return on sales for 2019 and compare the result to the industry average. Use the following financial data for Brenner Instruments Sales revenue.. Cost of goods...
-
Josh, Inc. is faced with the choice of either producing a newly designed product, XX-30, to stock in anticipation of demand or to customer order. The demand for the product is expected to be 5,000...
-
you have been called as an expert witness at a trial. Much of the evidence of the trail has been based on eyewitness accounts. For this discussion, present what you would relate to the jury regarding...
-
On January 1, 2018, Schmidt Industries purchased a new paint sprayer for $400,000. The salvage value after 5 years is estimated to be $100,000. The sprayer is depreciated by straight line...
-
Problem 51 of Chapter 6 presented a strategy for the synthesis of the amino acid (2S,3S)-3- hydroxyleucine, requiring as the starting material a specifi c stereoisomer of 2-bromo-3-hydroxy-4...
-
Show how you would synthesize each of the following molecules from an alkene of appropriate structure (your choice). Br H () (b) I (meso-4R,5S-isomer) H Br H Br Br H (d) (Racemate of 4R,5R and 4S,5S...
-
What is the unit product cost using variable costing? a. $50 per unit b. $55 per unit c. $70 per unit d. $90 per unit Donovan Company incurred the following costs while producing 500 units: direct...
-
Identify and briefly describe the principal types of (a) processing controls and (b) output controls.
-
Compare and contrast the ways in which consumers and organisations buy products and services.
-
What specific aspects of data processing are included in input controls?
-
What role does a data control group play in an EDP system?
-
What part should the sales function play when drawing up a detailed 12 months operational marketing plan for EMA?
-
The following table gives a number of Rockwell G hardness values that were measured on a single steel specimen. Compute average and standard deviation hardness values. 47.3 48.7 47.1 52.1 50.0 50.4...
-
Suppose you won a financial literacy competition and are given FJS10000 to invest, with the condition that investment can be done either in, i) Invest in Unit trust of Fiji or Invest in Fijian...
-
I-Butanol and chlorobenzene form a minimum-boiling azeotropic system. The mole fraction of I-butanol in the liquid (x) and vapour (y) phases at 1.000 atm is given below for a variety of boiling...
-
The following data have been obtained for the liquid-vapour equilibrium compositions of mixtures of nitrogen and oxygen at 100 kPa. T/K 77.3 78 80 82 84 86 88 90.2 X (O2) 0 10 34 54 70 82 92 100...
-
The table below gives the break and halt temperatures found in the cooling curves of two metals A and B. Construct a phase diagram consistent with the data of these curves. Label the regions of the...
-
Describe the shape of the distribution and look for any outliers. The data is skewed left with one unusually small outlier. The data is skewed right with one unusually large outlier. The data is...
-
Apex Fabricating wants to accumulate $ 8 2 0 , 0 0 0 for an expansion expected to begin in four years.If today Apex makes the first of equal quarterly payments into a fund earning 7 7 5 % compounded...
-
when reviewing income statments of an orangizaton, most annual statements provide ______ of income statement history

Study smarter with the SolutionInn App


