Give the expected product(s) of each of the following reactions. (a) (b) (c) (d) (e) (f) +
Question:
Give the expected product(s) of each of the following reactions.
(a)
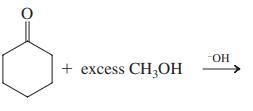
(b)
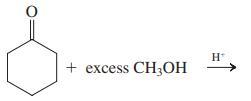
(c)
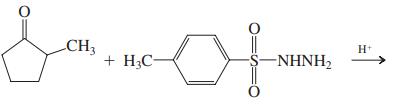
(d)
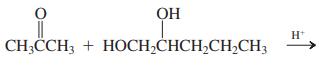
(e)
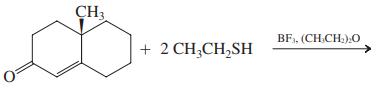
(f)
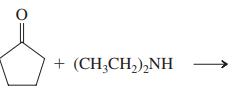
Transcribed Image Text:
+ excess CH,OH HO-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a ...View the full answer

Answered By

MANDWEEP BHUMIJ
I did my masters in Tezpur University with specialization in Organic chemistry. I cleared CSIR NET-JRF with all India ranking 74. I am currently working as Junior Research Fellowship in Central drug research institute Lucknow. I have some private tutoring experiences in multiple subjects.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the product of each of the following reactions: a. b. c. d. e. f. g. h. CH CH3 CH CH H2S04 CH,CH CHCH CH ,504 CH CH-CCH CH3 OH ' CH3O CH3OH 1. TsCl/pyridine 2. NaCEN CH3 OH (CH2CH2CH2hCuli CI
-
Give the product of each of the following reactions: a. b. c. d. e. f. g. h. hv CH3 H3C CH3 A DHHD H3C
-
Give the expected product(s) of each of the following reactions. 1. LDA, THF 2. BICH,COCH, H 1. LDA, THF 2. CH.CH Br, HMPA (a) CH,CCH,CH,CH3 (b) 0:
-
Rosa's employer has instituted a flexible benefits program. Rosa will use the plan to pay for her daughter's dental expenses and other medical expenses that are not covered by health insurance. Rosa...
-
1. What did you find interesting or noteworthy in reading this case? What can this situation teach us about effective leadership and/or world-class performance? 2. Why do people do this?
-
Explain why smaller and larger hedge funds generally have different risk and return objectives.
-
What stakeholder affected by their behavior was Gilead weighting very lightly when it decided what to tell the FDA about the medicines it wished to have permission to sell?
-
Accountants for Carlson, Inc. have assembled the following data for the year ended December 31, 2016: Prepare Carlsons statement of cash flows using the indirect method. Include an accompanying...
-
Lab: Structured Query Language (SQL) Queries with Data Manipulation Language (DML) Assignment Instructions LAB: STRUCTURED QUERY LANGUAGE (SQL) QUERIES WITH DATA MANIPULATION LANGUAGE (DML)...
-
1. Which process should VBB choose to produce?? 2. How much would VBP be willing to pay for the testing that is currently offered, for each batch?? 3. Would we be considered a perfect test, at twice...
-
Give the expected products of reaction of 4-acetylcyclohexene with each of the reagents in Problem 34. Data From Problem 34 (a) LiAlH 4 , (CH 3 CH 2 ) 2 O, then H + , H 2 O (b) CH 3 CH 2 MgBr, (CH 3...
-
Formulate detailed mechanisms for (a) the formation of the hemiacetal of acetaldehyde and methanol under both acid- and base-catalyzed conditions and (b) the formation of the intramolecular...
-
Nonexchange expenditures are the mirror image of nonexchange revenues. A state government provided several grants to school districts and local governments during its fiscal year ending August 31. 1....
-
Discuss the effect of the appropriation of Universal Monetary Announcing Benchmarks (IFRS) on the bookkeeping hones of multinational organizations. In your reply, consider the challenges, benefits,...
-
What are the specified tolerances that perishable stock should remain within?
-
Define the purpose of "Access Vector" and "Privileges Required" CVSS metric.
-
1. Do you think that current and possible future employers are crossing the line and invading an employee's privacy by viewing the employee's off-duty behavior? 2. Does the company you work for have...
-
What is the allowable depreciation for the computer in year one?
-
Tan Corporation purchased depreciable tangible personal property for $100,000 in 2012 and immediately expensed the entire cost under 179. In 2014, when the property was worth $80,000, Tan...
-
What are some of the possible sources of information about a company that could be used for determining the companys competitive stance?
-
Consider the species CH 3 , which has three normal carbonhydrogen bonds arid no other electrons on the carbon. (a) What is the charge of this species? (b) What is its geometry? (c) Discuss the...
-
Explain how the dipole moments of FCl (0.9 D) and 1Cl (0.7 D) can be so similar.
-
Chlorine is more electronegative than phosphorus. Predict the dipole moment of PCl5.
-
Read"Why Race Isn't as"Black"and White"as We Think"by Brent Staples. What can you find out about your own racial,religious,and cultural heritage? Search the Gateway library database: Ancestry Library...
-
Describe the classic steps for identifying an endocrine gland. Are these steps practical for identifying the sources of all the hormones we know of today? Explain.
-
Assuming Jeff has a mild chronic disease, and he has a private hospital health insurance. His salary before tax is $170,000. And assuming he has sold $30,000 of mutual fund units after he bought them...

Study smarter with the SolutionInn App


