How would you best classify the following reaction? (a) Wittig reaction; (b) Cyanohydrin formation; (c) Conjugate addition;
Question:
How would you best classify the following reaction?
(a) Wittig reaction;
(b) Cyanohydrin formation;
(c) Conjugate addition;
(d) Aldol addition.
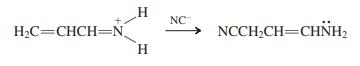
Transcribed Image Text:
H. NC -> H2C=CHCH=N H. NCCH;CH=CHNH,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
The correct option is C Conjugate addition These are basically ...View the full answer

Answered By

Vivek Kumar
Hi. I am Postgraduate in Chemistry (2012) with First class degree.
Expert in time bound exam assignment and quizzes. Did for many students
Seasoned online chemistry Tutor with 4 years of experience in tutoring across the globe.
Students loves my lessons and i get lots of appreciations and complements. I teach every part of chemistry ranging from Physical chemistry to Medicinal chemistry.
If you ask me speciality, i would say Organic chemistry, Spectroscopy (MS, NMR IR and UV etc) and Physical chemistry.
I am also tutoring maths.
I believe teaching in an art. Not every student is same and so the subject. Therefore it is the responsibility of Tutor to use that art to help students and make the subject interesting.
Thank You. Keep Smiling and keep learning
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
The Wohl degradation, an alternative to the Ruff degradation, is nearly the reverse of the Kiliani-Fischer synthesis. The aldose carbonyl group is converted to the oxime, which is dehydrated by...
-
Both the conjugate addition step and the intramolecular aldol condensation step can be carried out in one synthetic operation without isolating any of the intermediates along the way. For example,...
-
Tertiary amines undergo reversible conjugate addition to a,b-unsaturated ketones (see Chapter 18). This process is the basis for the Baylis-Hillman reaction, which is catalyzed by tertiary amines,...
-
Two franchising experts recently debated the issue of whether new college graduates should consider franchising as a pathway to entrepreneurship. Jeff Elgin said recent college graduates are not...
-
In 1994, a federal law was enacted giving victims of gender-motivated violence (i.e., sexual assault) the right to sue for civil damages. Do you think such a law is constitutional? See the case...
-
In Exercises 107118, begin by graphing the cube root function, f(x) = 3x. Then use transformations of this graph to graph the given function. g(x) = x + 2
-
The statement "Amounts due to vendors should be accurately and promptly classified, summarized, and reported" might serve as a control objective in which of the following transaction cycles? (a)...
-
The unadjusted trial balance of Arlington Air Purification System at December 31, 2014, and the data needed for the adjustments follow. Adjustment data at December 31 follow: a. On December 15,...
-
While Netflix's distribution network was larger than rivals, do you believe it would have been realistic for competitors to combat the problem and mimic Netflix's DC network? Do you believe Netflix's...
-
Marshall defined an equilibrium price as one at which the quantity demanded equals the quantity supplied. a. Using the data provided in problem 1.1, show that P = 3 is the equilibrium price in the...
-
When 3-methyl-1,3-diphenyl-2-butanone is treated with excess D2O in the presence of catalytic acid, some of its hydrogens are replaced by deuterium. How many? (a) One; (b) Two; (c) Three; (d) Six;...
-
The aqueous hydroxide-promoted reaction of the compound shown in the margin with (CH 3 ) 3 CCHO yields exclusively one compound. Which one is it? (a) (b) (c) (d) H;CC -CH;
-
Refer to Problem 2-36 in Chapter 2 for The Hughes Tools Company. a. Prepare journal entries for each of the transactions and adjustments listed in the problem. b. Prepare the necessary T accounts and...
-
Internal control is strengthened when the quantity of merchandise or- dered is omitted from the copy of the purchase order sent to the a. Department that initiated the requisition. b. Receiving...
-
The NCHS keeps statistics on ____ . a. The licensing information on all healthcare providers in the 50 states b. Cancer and other deadly diseases in the 50 states and the US-owned territories c....
-
Negative confirmation of accounts receivable is less effective than posi- tive confirmation of accounts receivable because a. A majority of recipients usually lack the willingness to respond...
-
In a properly designed accounts payable system, a voucher is prepared after the invoice, purchase order, requisition, and receiving report are verified. The next step in the system is a. Cancellation...
-
Which user of statistics has the primary job of supporting terminally ill patients and their families? a. Home health agencies b. Nursing facilities c. Hospice d. MCOs
-
Describe the difference between criticism and coaching feedback.
-
1A. If the researcher is concerned about the number of variables, the nature of the analysis, and completion rates, then, he/she is at which stage of the sampling design process (Figure 11.1 in the...
-
The redox reactions of quinones have been the subject of many studies over the years and they continue to be of interest to electrochemists. In a study of methane (1,I-dimethyl-3,5-cyclohexanedione)...
-
Although the attractive van der Waals interaction between individual molecules varies as R-6, the interaction of a molecule with a nearby solid (a homogeneous collection of molecules) varies as R-3,...
-
If the dependence of the chemical potential of the gas on the extent of surface coverage is known, the Gibbs adsorption isotherm, eqn 19.50, can be integrated to give a relation between Va and p, as...
-
(Computing the expected rate of return and risk) After a tumultuous period in the stock market, Logan Morgan is considering an investment in one of two portfolios. Given the information that follows,...
-
CMS Corporation's balance sheet as of today is as follows: Long-term debt (bonds, at par) Preferred stock Common stock ($10 par) Retained earnings Total debt and equity $10,000,000 2,000,000...
-
content area top Part 1 An exercise machine with an original price of $ 690 is on sale at 10 % off. a. What is the discount amount? b. What is the exercise machine's sale price? Question content area...

Study smarter with the SolutionInn App


