In this transformation, what is the best structure for A? (a) BrCH 2 CH 2 CH(CH 3
Question:
In this transformation,
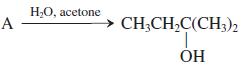
what is the best structure for A?
(a) BrCH2CH2CH(CH3)2
(b)
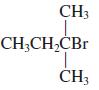
(c)
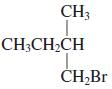
(d)
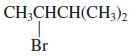
Transcribed Image Text:
Н-О, асetone A CH;CH,C(CH3)2 ÓH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
In this reaction SN 1 mechanism is follow If we consider the A as 1 h...View the full answer

Answered By

Vivek Patel
I have teaching experience for 1 year at Nalanda Higher secondary complex.
I have taken Chemistry class of standard 11th and 12th there.
it was very great experience for me...
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
In this transformation, what is the best structure for "A"? (a) (b) (c) (d) , 1. LiAlH, dry ether 2. H., H,O (work-up) CH-CH CH2CH CH2CH3 CH2CH -, - -,
-
A certain hydrocarbon containing eight carbons was found to have two degrees of un-saturation but no absorption bands in the IR spectrum at 1640 cm-1. The best structure for this compound is a. b. c....
-
Give the IUPAC names of the following alkanes. (a) CH3C(CH3)2CH(CH)CH3)CH2CH2CH(CH3)2 (b) (c) (d) (e) (f) (g) (h) CH,CH CHCH CH, CH CH CH,CHCH CH,CHCH CH,CH CH,CH, CH, CH,CH, CH,CH,CH, C(CH,CH),...
-
The adjusted trial balances of Rosa Corporation at August 31, 2014, and August 31, 2013, include these amounts (in millions): Rosa completed these transactions (in millions) during the year ended...
-
Describe the resources that are required to work on the procurement proposal selection committee.
-
Referring to the facts in question 5 above, what is the material quantity variance? a. $5,000 unfavorable b. $5,000 favorable c. $2,000 favorable d. $2,000 unfavorable
-
Panther Racing, LLC, is involved in competitive racing, including the development, maintenance, and operation of a racing team in the IndyCar series. Beginning in 2008, the National Guard annually...
-
Instant Dinners, Inc. (IDI), is an established manufacturer of microwavable frozen foods. Leland Forrest is a member of the planning and analysis staff. Forrest has been asked by Bill Rolland, chief...
-
Crystal Corporation earned net income of $ 9 0 0 , 0 0 0 in 2 0 2 3 . It has a complex capital structure as follows: 1 2 , 0 0 0 shares outstanding of 8 % , $ 1 0 0 preferred stock, and 1 7 0 , 0...
-
"Part 1: The Performance Lawn Equipment database contains data needed to develop a pro forma income statement. Dealers selling PLE products all receive 18% of sales revenue for their part of doing...
-
Consider the general substitution-elimination reactions of the bromoalkanes. How do the reaction mechanisms and product formation differ when the structure of the substrate and reaction conditions...
-
Which of the following isomeric carbocations is the most stable? CH,+ CH3 () (b) -CH3 CH3 () (d)
-
In Problems 41 52, the graph of an equation is given. (a) Find the intercepts. (b) Indicate whether the graph is symmetric with respect to the x-axis, the y-axis, the origin, or none of these....
-
GIVE COMPREHENSIVE EXAMPLES OF HOW INACCURATE,INCOMPLETE,OR OUTDATED DATA LEAD TO MEDICAL ERRORS,DELAYS IN TREATMENT OR OTHER PROBLEMS
-
Can the concept of "total institutions," as conceptualized by Goffman, provide insights into the ways in which enclosed, all-encompassing environments such as prisons, military barracks, and...
-
There are many professional groups to which an insurance professional can belong, on both the local and national level. Give some examples of these local and national level insurance professional.
-
Manufacturers Southern leased high-tech electronic equipment from Edison Leasing on January 1, 2018. Edison purchased the equipment from International Machines at a cost of $117,772. (FV of $1, PV of...
-
Discuss 2 benefits of recording these transactions as a liability versus paying them immediately
-
On January 1, 2011, Alpha acquired 80 percent of Delta. Of Delta's total business fair value, $125,000 was allocated to copyrights with a 20-year remaining life. Subsequently, on January 1, 2012,...
-
Kenneth Hubbard has prepared the following list of statements about managerial accounting and financial accounting. 1. Financial accounting focuses on providing information to internal users. 2....
-
Explain how colligative properties are used to determine molar mass.
-
The fugacity coefficient of a certain gas at 290 K and 2.1 MPa is 0.68. Calculate the difference of its molar Gibbs energy from that of a perfect gas in the same state.
-
Explain what is meant by a regular solution.
-
You are given the following ODE: y+3y+2y+ y = 3x 1. Derive the frequency response function G(jw) of this ODE. 2. Calculate the magnitude of G(jw). 3. Calculate the phase of G(jw). 4. Calculate the...
-
Calculate the moment of inertia in terms of a and b according to the x-axis of the area in the figure -a- y=k(x-a)3 20 b
-
A premium annual - pay bond pays a $ 8 2 coupon, has a yield to maturity of 5 . 1 7 % , and is priced at $ 1 , 0 9 3 . 9 8 . How many years till the bond matures? Answer in years to at least two...

Study smarter with the SolutionInn App


