Propose a structure for a compound that displays the spectroscopic data that follow. The molecular ion in
Question:
Propose a structure for a compound that displays the spectroscopic data that follow. The molecular ion in the mass spectrum appears at m/z = 116. IR v˜ = 1710 (s) and 3000 (s, broad) cm-1. 1H NMR: δ = 0.94 (t, J 5 7.0 Hz, 6 H), 1.59 (m, 4 H), 2.36 (quin, J 5 7.0 Hz, 1 H), and 12.04 (broad s, 1 H) ppm; 13C NMR: 11.7, 24.7, 48.7, and 183.0 ppm.
Transcribed Image Text:
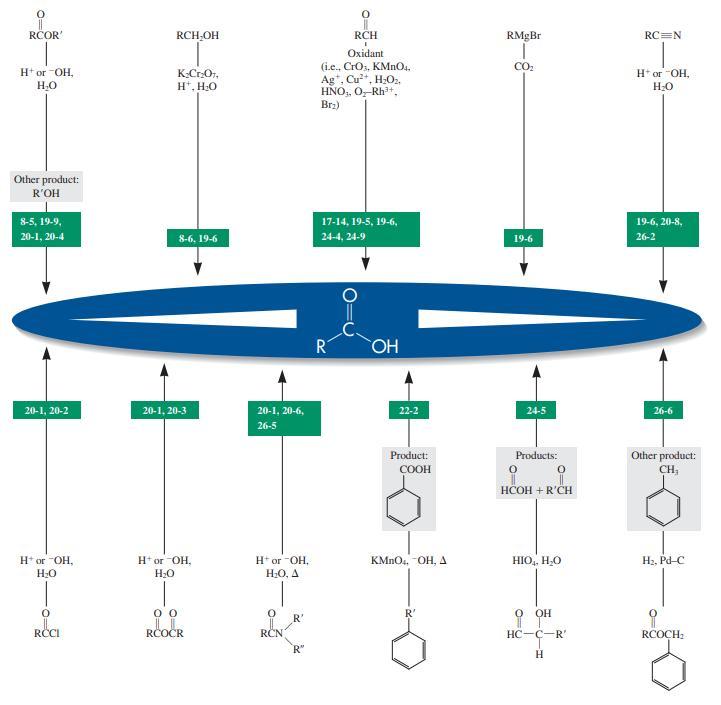
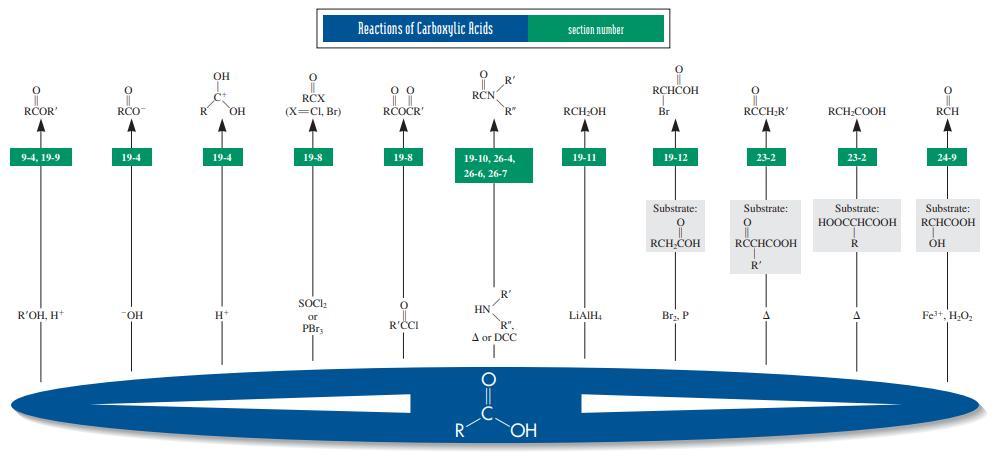
RCOR' RCH,OH RCH RMgBr RC=N Oxidant (i.e., CrOs, KMNO4. Ag*, Cu", H;O:. HNO,, O,-Rh'+. Brz) CO2 Htor OH, H* or "OH, K-CrOr, H*, H;0 Other product: R'OH 8-5, 19-9, 17-14, 19-5, 19-6, 19-6, 20-8, 20-1, 20-4 8-6, 19-6 24-4, 24-9 19-6 26-2 R HO 20-1, 20-2 20-1, 20-6, 22-2 24-5 20-1, 20-3 26-6 26-5 Other product: CH Product: Products: ÇOOH HCOH + R'CH H* or -OH, H or OH, H* or OH, H:0, A KMNO.. OH, A HIO,, HO H2, Pd-C R' он RCI RČOČR RCN HC-C-R' RCOCH: R"
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
Given m2 116 molecular ion The IR data corresponding to 1710s cm 1 suggests carb...View the full answer

Answered By

Abhishek rawat
I am very hardworking and believe in smartwork of students and solutioinn provide me the platform to interact with students and I love to work in this platform I have already worked in many online platforms and want to continue to do same and help children to achieve there goals in life.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Compound M has the molecular formula C9H12. The 1H NMR spectrum of M is given in Fig. 14.29 and the IR spectrum in Fig. 14.30. Propose a structure for M.
-
Compound Z (C5H10O) decolorizes bromine in carbon tetrachloride. The IR spectrum of Z shows a broad peak in the 3200-3600-cm-1 region. The 300-MHz 1H NMR spectrum of Z is given in Fig. 21.3. Propose...
-
Following are spectroscopic and other data for several compounds. Propose a structure for each of them. (a) Molecular formula = C 6 H 4 Br 2 . 1 H NMR spectrum A. 13 C NMR: 3 peaks. IR: v = 745 (s,...
-
X = {a,b,c,d. e, f} olmak zere (X.) ksmi sral kmesinin grafii aada verilmitir. (a) (X.) ksmi sral kmesinin maksimal, minimal, minimum ve maksimum elemanlarn belirtiniz. (b) (X.) kismi sral kmesinin Y...
-
Now, suppose that U.S. policymakers do not actually care about pollution, but merely about real purchasing power of U.S. incomes. What is the effect of an increase in on the terms of trade, and on...
-
The behavior of invertebrates such as Aplysia is totally: (a) controlled by genes (b) learned (c) reflexive (d) none of the above
-
Consider the following cash flow diagram. What is the value of \(X\) if the internal rate of return is 15 percent? a. \(\$ 246\) b. \(\$ 255\) c. \(\$ 281\) d. \(\$ 290\) 0 $400 X X 1 2
-
Almer Company operates two factories. The company applies factory overhead to jobs on the basis of machine hours in Factory 1 and on the basis of direct labor hours in Factory 2. Estimated factory...
-
Write a Java program to Connect Java Application with Oracle database. Please, use the keyboard and support your answers with a screenshot
-
Summarized versions of Calabasa Corporations financial statements for two recent years are as follows. Requirement 1. Complete Calabasa Corporations financial statements by determining the missing...
-
Rank each of the following groups of organic compounds in order of decreasing acidity. (a) (b) (c) (d) (e) CH;CH,CO,H, CH;CCH,OH, CH;CH,CH,OH
-
(a) An unknown compound A has the formula C 7 H 12 O 2 and infrared spectrum A (p. 917). To which class does this compound belong? (b) Use the other spectra (NMR-B, p. 917, and F, p. 919; IR-D, E,...
-
In the short run the perfect competitor may make a (n) or take a (n) ____________-; in the long run the perfect competitor will __________.
-
Based on economists' forecasts and analysis, one-year Treasury bill rates and liquidity premiums for the next four years are expected to be as follows: R 1 = 5.75% E ( r 2 ) = 6.85% L 2 = .55% E ( r...
-
If an insurance company doesn't quickly approve a procedure and release funds, what can the practice do to expedite and improve cash flow?
-
What are the intricacies of network bootstrapping protocols like Preboot Execution Environment (PXE) and Dynamic Host Configuration Protocol (DHCP), and how do they function in provisioning operating...
-
A company is considering a 3-year project that requires an initial installed equipment cost of $14,000. The project engineer has estimated that the operating cash flows will be $4,000 in year 1,...
-
Your degree program requires a total 127 credit hours. You have taken 37 credit hours so far. So, you have to take more 90 credits hours to complete your degree. If you take 27 credit hours per...
-
Puce is a corporation that buys and sells financial assets. It purchases notes receivable from manufacturers that need cash immediately and cannot wait to collect the notes. Puce pays about 88% of...
-
As indicated by mutual fund flows, investors tend to beat the market seek safety invest in last year's winner invest in last years loser
-
Provide the values for the missing K a or p K a in the following examples: (a) p K a = 4; K a =? (b) K a = 1 x 10 16 ; p p K a =? (c) p K a = 38; K a =? (d) K a = 1 x 10 6 ; p K a =?
-
Indicate whether these compounds are weaker or stronger acids than water (the K a for water is 1.8 x 10 ?16 ; the p K a is 15.74): (K=100) a) HCIO4 c) HOCOH (PK = 6.35) b) HCCH (pK = 25) d) CH3CH...
-
Indicate whether these species are weaker or stronger bases than hydroxide ion. The Ka is or pKa values are for the conjugate acids. a) :NH, (K = 10-38) c) NH, (PK, = 9.24) b) CHCHCH d) :CI: (pK =...
-
Saskatewan Can Company manufactures recyclable soft-drink cans. A unit of production is a case of 12 dozen cans. The following standards have been set by the production-engineering staff and the...
-
Assume that you hold 100 Vodacom shares which you intend to sell in a month's time. To protect yourself from future price risks, you then decided to construct a covered call strategy using a call...
-
The four people below have the following investments. Invested Amount Interest Rate Compounding Jerry Elaine $ 11,900 12% Quarterly 14,900 10 Semiannually 21,900 6 Annually 17,900 8 Annually George...

Study smarter with the SolutionInn App


