Reaction review. Without consulting the Reaction Road Map on pp. 816 817, suggest reagents to convert each
Question:
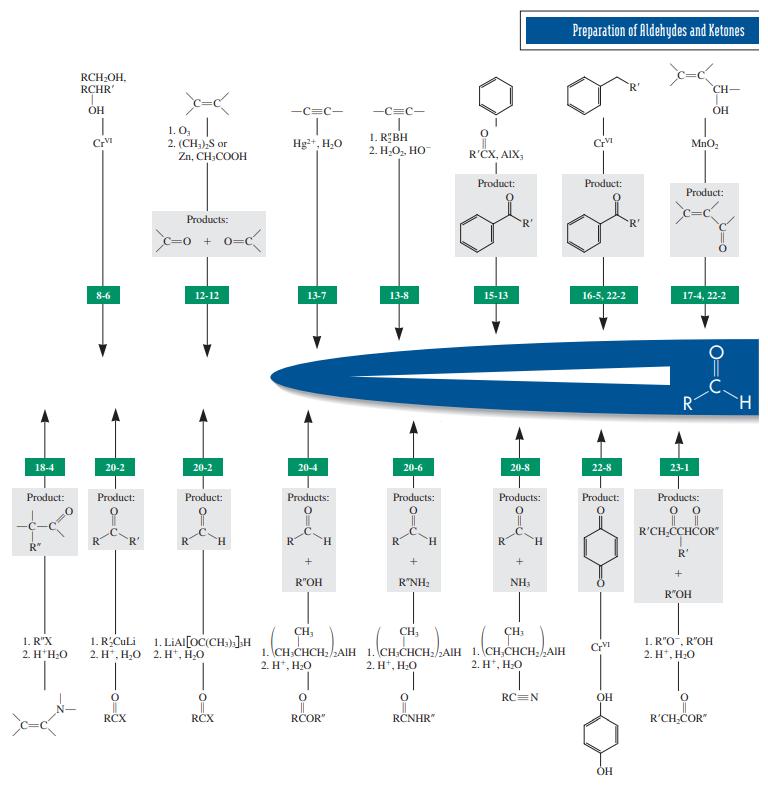
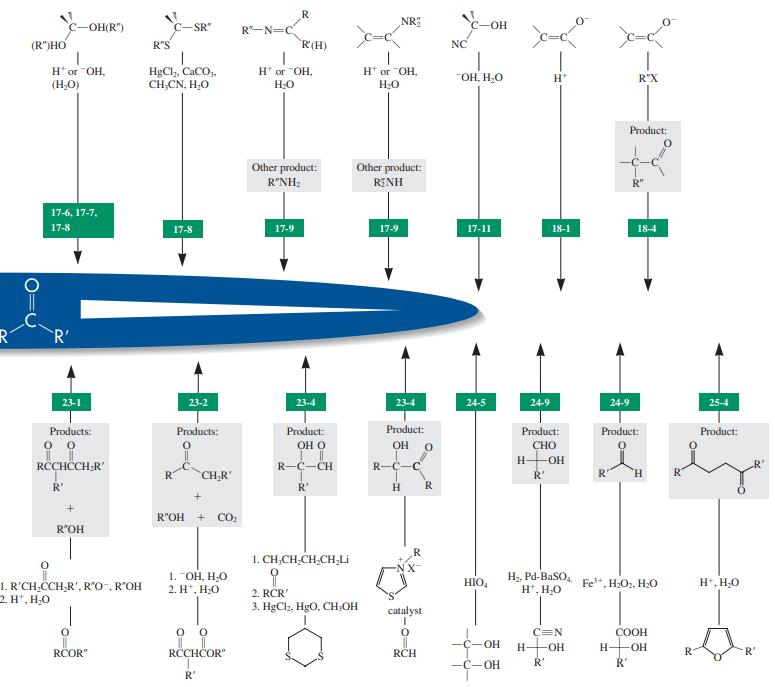
Reaction review. Without consulting the Reaction Road Map on pp. 816 –817, suggest reagents to convert each of the starting materials below into 3-hexanone.
(a)
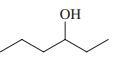
(b)
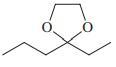
(c)
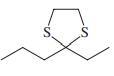
(d)
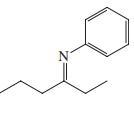
(e)
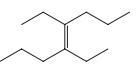
(f)
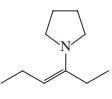
(g)

(h)
page 816-817
Transcribed Image Text:
ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 20% (5 reviews)
a b c d e f g h S ...View the full answer

Answered By

Jatin Verma
Life is a chemical reaction, it only requires balancing! To balance your minds with my more than six years of experience I Jatin Verma here for all you better things for better living through chemistry and also to solve mysteries in chemistry I as your chemistry educator without even touching your arm it will set fire to your minds.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Reaction review. Without consulting the Reaction Road Map on p. 721, suggest a reagent to convert benzene into each of the following compounds. C(CH3)3 NO2 CH3 CH,CH; SO,H Br (e) (f) (h)
-
Reaction review II. Without consulting the Reaction Road Map on p. 818, suggest reagents to convert cyclohexanone into each of the compounds below. -5 -8 - OH CH2CH3 CN (b) () (d) N(CH,CH,), () (f)...
-
Reaction review. Without consulting the Reaction Road Map on p. 598, suggest reagents to convert a general alkyne RCCH into each of the following types of compounds. Br Br I| | () R- R Br () C=C (b)...
-
An analysis of spending by a sample of credit card bank cardholders shows that spending by cardholders in January (Jan) is related to their spending in December ( Dec): The assumptions and conditions...
-
What role did the Eighth Amendment play in the Court's decision in this case?
-
Identify and explain group dynamic biases that might affect a board of directors.
-
Petitioner Curtis Flowers has been tried six separate times for the murder of four employees of a Mississippi furniture store. Flowers is black; three of the four victims were white. At the first two...
-
John Fuji (age 37) moved from California to Washington in December 2013. He lives at 468 Cameo Street, Yakima, WA 98901. Johns Social Security number is 571785974 and he is single. His earnings and...
-
What is Data Manipulation Language in SQL ? What are the Benefits of DML? What is DML command with example?
-
Three years ago, the Oak Street TCBY bought a frozen yogurt machine for $11,200. A salesman has just suggested to the TCBY manager that she replace the machine with a new, $13,500 machine. The...
-
Following are spectroscopic and analytical characteristics for an unknown compound. Propose a structure. Empirical formula: C 8 H 16 O. 1 H NMR: = 0.90 (t, 3 H), 1.0 1.6 (m, 8 H), 2.05 (s, 3 H),...
-
Indicate which reagent or combination of reagents is best suited for each of the following reactions. (a) (b) (c) (d) (e) (f) OH
-
Find an equation of the osculating circle at the point indicated or indicate that none exists. r(t) = (1 si n t, 1 2 cos t), t =
-
Does the possession of a core competence guarantee success? If yes, explain why. If no, explain why.?
-
Briefly describe the connection between Income Statement and Balance Sheet.
-
Calculate break-even volume in units (rounded): Fixed costs $125,000 Price per unit 5.75 Variable costs per unit 3.75
-
CSU Corporation began operations on January 1, 2017. The following information is available for CSU on December 31, 2017: Accounts receivable 1,800 Retained earnings ? Supplies expense 200 Accounts...
-
Consider the following code fragment. What do we need to enforce on function myFun() so that classes BBB and CCC can be used polymorphically? class AAA {my Fun() {}; }; class BBB: public AAA {...};...
-
Kelly recently was promoted and received a substantial raise. She talks to her tax adviser about the potential tax ramifications. After making some projections, her adviser "welcomes her to the AMT...
-
CdF2 (s) Cd+ (aq) + 2 F- (aq) 1. A saturated solution of CdF2 is prepared. The equilibrium in the solution is represented above. In the solution [Cd+] eq = 0.0585 M and [F-] eq = 0.117 M. a....
-
Explain whether the bonds in these compounds would be ionic or covalent and show Lewis structures for them: (a) KC1 (b) NC13 (c) NaCN (d) KOH
-
What is the formula for the simplest neutral compound formed from P and H? Show a Lewis structure for this compound and predict its shape.
-
Show Lewis structures for these compounds: (a) CH5N (b) C 2 H 5 Cl (c) N 2 (d) CH 2 S (e) C 2 H 3 F (f) CH 4 S
-
Georgias employee will match her contributions into a retirement plan up to 5% of her 40,000 annual salary. in other words, the employee will put $1 into her retirement plan for every $1 Georgia puts...
-
Stock in Country Road Industries has a beta of 1.09. The market risk premium is 7.5 percent, and T-bills are currently yielding 3.5 percent. The company's most recent dividend was $1.7 per share, and...
-
Festival Dancing and Fitness In this activity, you will be provided with a review on the implication of dancing activity to your fitness by way of determining your range of Target Heart Rate. Let us...

Study smarter with the SolutionInn App


