Select the acid with the highest K a (i.e., lowest pKa). (a) H 3 CCO 2 H
Question:
Select the acid with the highest Ka (i.e., lowest pKa).
(a) H3CCO2H
(b)
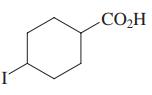
(c)
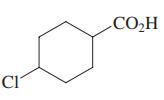
(d) (d) Cl2CHCO2H
Transcribed Image Text:
CO,H I
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Calculate Ka for each of the following acids, given its pKa. Rank the compounds in order of decreasing acidity. (a) Aspirin: pKa = 3.48 (b) Vitamin C (ascorbic acid): pKa = 4.17 (c) Formic acid...
-
Acid HA has pKa = 20; acid HB has pKa = 10. (a) Which is the stronger acid? (b) Will an acid-base reaction with an equilibrium lying to the right take place if Na+A- s added to HB? Explain your...
-
(a) An acid (HA) has Ka = 10-7. What is its pKa? (b) Another acid (HB) has Ka = 5.What is its pKa? (c) Which is the stronger acid?
-
Suppose that the owner of the restaurant in Problem 2.91 also wants to study the demand for dessert during the same time period. She decides that in addition to studying whether a dessert was...
-
Consider a country that is a net importer of oranges, in a partial-equilibrium comparative advantage model of the same type as the U.S.-ROW sugar model in the text. This is a country that does...
-
List the eight major groupings of culture actions and provide some examples for each group.
-
Reconsider Parts (a) through (f) of Problem 2. For each "true" statement, develop a mathematical proof based on the time value of money factor equations from Table 2.6 in Chapter 2. Data from problem...
-
Fran Omiyale started her own consulting firm, Omiyale Consulting, on May 1, 2012. The trial balance at May 31 is as follows. In addition to those accounts listed on the trial balance, the chart of...
-
3. A bag of mixed nuts contains almonds and hazelnuts. There are (6x+13) nuts in this particular bag, and (3x-7) of these are hazelnuts. Which expression represents the number of almonds in the bag?...
-
Verify the values given in Table 5.3 for NV, NE, and ND for a partial reboiler and a totalcondenser. Ng. Independent Relationships Ny, Total Number of No. Degrees of Freedom Element or Unit Name...
-
Rank each of the following groups of organic compounds in order of decreasing acidity. (a) (b) (c) (d) (e) CH;CH,CO,H, CH;CCH,OH, CH;CH,CH,OH
-
The acid whose structure is shown in the margin can best be prepared via one of these sequences. Which one? (a) (b) (c) (d) HO,C C(CH3)3
-
What is a minimum resale price maintenance agreement? How might it reduce competition and when might it be acceptable?
-
A local business plans on advertising their new product by purchasing advertisements on the radio and on TV. The business plans to purchase at least 60 total ads and they want to have at least three...
-
CCC Inc. has a target capital structure of 60 percent equity and 40 percent debt. The flotation costs for equity issues are 8 percent of the amount raised; the flotation costs for debt issues are 3...
-
Read this New York Times article linked on the assignment page and share your thoughts. Failed by Law and Courts, Troops Come Home to Repossessions By JESSICA SILVER-GREENBERG and MICHAEL CORKERY...
-
A firm is considering a project that will generate perpertual after-tax cash flows of 25,000 per year beginning next year. The project has the same risk as the firm's overall operations. Equity cost...
-
Van Doren Housing expects to have sales this year of $15 million under its current credit policy. The present terms are net 30; the days sales outstanding (DSO) is 60 days; and the bad debt loss...
-
Albert owns 100 acres of land on which he grows spruce Christmas trees. His adjusted basis for the land is $100,000. He receives condemnation proceeds of $10,000 when the city's new beltway takes 5...
-
Solve for the equilibria of the following discrete-time dynamical systems Pr pt+1 = Pr+2.0(I-Pr)
-
Consider the two stereo isomers of 1, 4-dimethyl cyclohexane: (a) Explain whether each methyl is axial or equatorial in the conformations of the cis-isomer. (b) Explain whether each methyl is axial...
-
Consider the two stereo isomers of 3-isoproplcyclohexanol. (a) Which is the more stable conformation of each stereo isomer? (b) Which is the more stable stereo isomer by how much?
-
For thee compounds indicated whether the substituents are cis or trans whether they are axial or equatorial, whether the conformation shown or the other chair conformation is more stable, and whether...
-
The first page of the My Autobiographical Playlist Document is for you to share your album cover , which can be drawn or created digitally. The remaining pages of the My Autobiographical Playlist...
-
Georgina is almost ready to graduate from high school. She has earned good grades and has participated in several science competitions while in high school. Her parents have encouraged her to learn...
-
PowerPoint is a visual aid for many speakers. Discuss some points to remember when adding text to a PowerPoint presentation. How do they help make the experience better for the audience and the...

Study smarter with the SolutionInn App



