The following syntheses are proposed for amines. In each case, indicate whether the synthesis will work well,
Question:
The following syntheses are proposed for amines. In each case, indicate whether the synthesis will work well, poorly, or not at all. If a synthesis will not work well, explain why
(a)
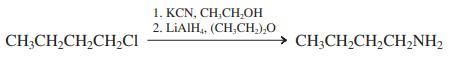
(b)
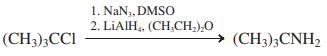
(c)
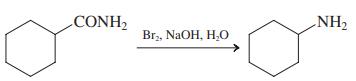
(d)
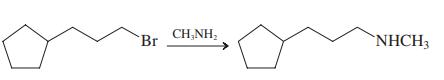
(e)
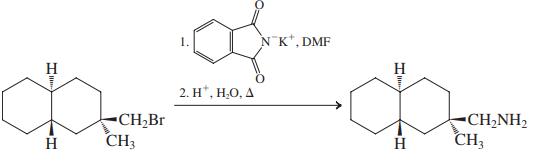
(f)
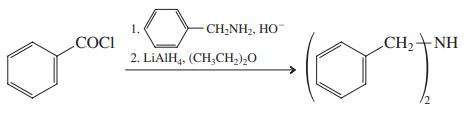
(g)
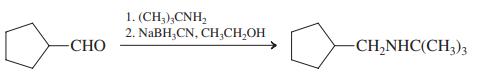
(h)
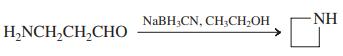
(i)
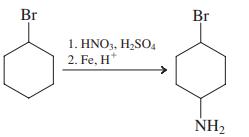
(j)
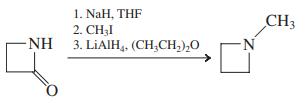
Transcribed Image Text:
1. KCN, CH,CH,ОН 2. LIAIH, (CH,CH,),O CH;CH2CH,CH,CI → CH;CH,CH,CH,NH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (11 reviews)
ANot at allThis process adds a carbonCN group making 1...View the full answer

Answered By

Samee Ullah
Algebra, Linear algebra, calculus, accounting, marketing, statistics, programming, real estate, writing, human resource management, business communication, Engineering: civil, chemical, electrical, mechanical, aerospace, building
Linguistics: sociolinguistics, applied linguistics, music, social sciences, biology, chemistry: all types, Thermodynamics, mechanics, modern physics, quantum physics, metaphysics, biology.
Feel free to contact us for all these subjects,; for quality, and best responses. Thankyou
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Name the following compounds: a. b. c. d. e. f. g. h. i. j. CH2CH3 CH,CH-CHCH-CH-CH-COH CH.CH,COCH.CH.CH. CH CH,CH CH C N CH.CH COCCH.CH CH3CH2COCCH2CH CH3CH2CH2CN(CH3)2 CH3CH2CH CH2CC COCCH CH CHCH...
-
Predict the products of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) OH CI CH NH2 Ph-C-CI+ NH2 0 + (D LiAIH (2) H20 (2) H,0 -. O (I) excess PhMgBr (2) H,o ( CH Mgl (2)...
-
Give the product of each of the following reactions a. b. c. d. e. f. g. h. i. j. CHCH CH,OH -1. methanesulfonyl chloride 2. CH CO CH,CH,CH,CH,OH + PBr, Pyridine 1. p-toluenesulfonyl chloride CH:CHCH...
-
Coronado Industries has adopted the following production budget for the first 4 months of 2022. Each unit requires 4 pounds of raw materials costing $6 per pound. On December 31, 2021, the ending raw...
-
How does the FE curve relate to the concept of external balance?
-
In Problems 7992, find the real solutions, if any, of each equation. Use any method. 10x 2 - 19x - 15 = 0
-
Consider a publicly held company whose products you are familiar with. Some examples might include: Access the companys public website and search for its most recent annual report. (Some companies...
-
Aquatic Biotechnology Inc. (ABI) is a medium-sized, public company operating an aquaculture business in eastern Canada. The company has been in operation since the mid-1990s, and during the latter...
-
1. A jeepney driver, Rod, entered into an agreement with Francisca that he will give her a ride from Baguio to Manila and that any accident that may occur will not make Rod liable. Is the contract...
-
Kraft Foods successfully introduced DiGiorno Pizza into the marketplace in 1996, with first year sales of $120 million, followed by $200 million in sales in 1997. It was neither luck nor coincidence...
-
Without consulting the Reaction Road Map on page 1009, suggest reagents that convert each of the following starting materials into the indicated product: (a) (chloromethyl)- benzene (benzyl chloride)...
-
For each synthesis in Problem 39 that does not work well, propose an alternative synthesis of the final amine, starting either with the same material or with a material of similar structure and...
-
A light ray in dense flint glass with an index of refraction of 1.655 is incident on the glass surface. An unknown liquid condenses on the surface of the glass. Total internal reflection on the...
-
Answer these questions about the history of auditing. a. Describe a method of auditing that seemed to be prevalent in ancient Egypt and Babylonia. b. Describe the audit of public accounts in ancient...
-
In what way did the Hochfelder case put limitations on auditor liability under Section 10 (b) of the Securities Exchange Act of 1934 ?
-
Given a unitary representation of a group on the vector space $V$, in which a scalar product $\langle\ldots \mid \ldotsangle$ is defined, and given a submodule $W$, and its orthogonal complement...
-
Explain the significant differences between the standards of liability under Section 11 (a) of the Securities Act of 1933 and Section 18 (a) of the Securities Exchange Act of 1934.
-
Explain why each of the following audits would be classified as (1) financial statement, (2) operational, or (3) compliance. a. Audit of the contract costs of building jet fighter planes. b. Audit by...
-
Explain why organizations need coordination across departments and hierarchical levels, and describe mechanisms for achieving coordination.
-
On average there are four traffic accidents in a city during one hour of rush-hour traffic. Use the Poisson distribution to calculate the probability that in one such hour there arc (a) No accidents...
-
Show the products of thesereactions: CH O PhS Na b) Ph,P + CH;CH;CH,Br benzene a) ELOH + CH;I c) CH;CH,CH,CH,S d) NASCH,CH2SNA + BRCH,CH,Br CH
-
Show the products of thesereactions: C, CH3 H,SO4 b) Co + CH,O a) + CH,OH CH, CH, 1) LIAIH4, ether c) 2) ,"
-
Show the products of thesereactions: Br S t-BUOH a) CH,CH,CH,CH, + t-BUOK + NaOCH,CH, b) Br d) CH;CH,CCH,CH3 + H,CH, CH-O . + CH;ONa CH
-
Which accounting standards are used by kogan.com and kathmandu retailer in 2023 by their annual report
-
Assessing risk and return ( You are required to recommend a two-asset portfolio from the following four assets, assuming equal investment in each asset that will minimise risk. ASX Code Name JBH...
-
Jack asked Jill to marry him, and she has accepted under one condition: Jack must buy her a new $ 3 5 0 , 0 0 0 Rolls - Royce Phantom. Jack currently has 3 6 , 0 4 0 that he may invest. He has found...

Study smarter with the SolutionInn App


