The mass spectra of both of the compounds described in Problem 61 show two molecular ion peaks,
Question:
The mass spectra of both of the compounds described in Problem 61 show two molecular ion peaks, two mass units apart, in an intensity ratio of about 3 : 1. Explain.
In Problem 61
Transcribed Image Text:
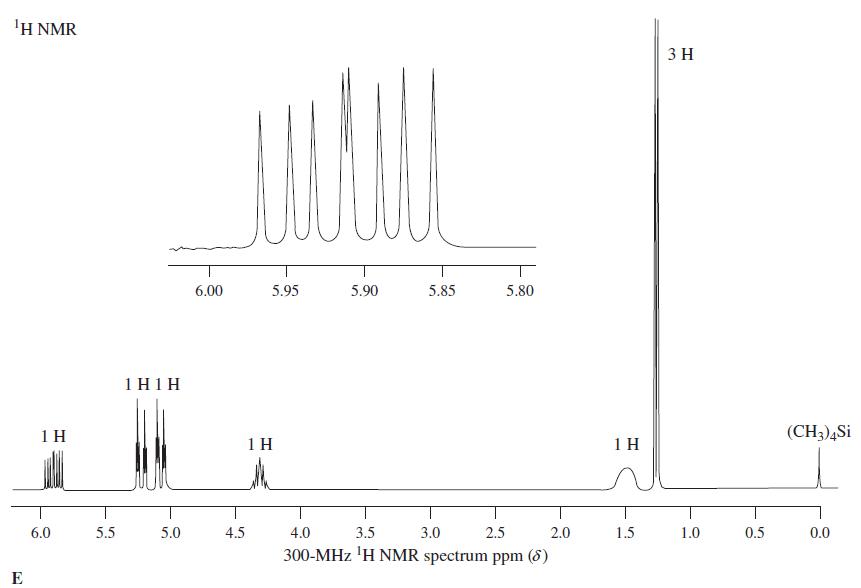
1Η ΝMR 3 H 6.00 5.95 5.90 5.85 5.80 1 H1H 1 H 1 H 1 H (CH3)4Si 6.0 5.5 5.0 4.5 4.0 3.5 3.0 2.5 2.0 1.5 1.0 0.5 0.0 300-MHz 'H NMR spectrum ppm (8) E
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
This suggests that the compounds have two different isotopes present in them Isotopes are atoms of t...View the full answer

Answered By

Ritul Choudhary
I am excited to introduce myself as a highly motivated and dedicated individual, who has a passion for the subject of Life Sciences. I completed my Higher Secondary education in the Montessori method of training, which has helped me to develop a unique perspective on teaching and learning. I am confident that my knowledge and skills, along with my Montessori background, make me an ideal candidate for the position. I have completed the first year of my Bachelor's degree in Life Science with a strong academic record, and I am a quick learner and have a strong ability to adapt to different situations. I am also comfortable using technology and different teaching methods to enhance the learning experience. I am looking forward to the opportunity to share my knowledge and help students achieve their full potential.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
The compounds described in Problem 27 have very different ultraviolet spectra. One has max () = 232(13,000) and 308(1450) nm, whereas the other has max () = 272(35,000) nm and a weaker absorption...
-
The 1H NMR spectra of two compounds with molecular formula C11H16 are shown here. Identify the compounds. a. b. 4 (ppm) 0 10 (ppm) frequency
-
The mass spectra of 1-methoxybutane, 2-methoxybutane, and 2-methoxy-2-methylpropane are shown in Figure 13.7. Match the compounds with the spectra. 100 73 80 S 60 57 20 0 10 20 30 40 50 60 70 80 90...
-
Which of these situations would require auditors to append an emphasis- of- matter paragraph about consistency to an otherwise unmodified opinion? a. Entity changed its estimated allowance for...
-
What is the purpose of the stakeholder communication matrix? Describe its contents.
-
Kelly Sellers is really fed up with his departments performance. He knows that his people have a very boring job, and the way the technological process is set up leaves little latitude for what he...
-
Hemlock Semiconductor Operations, LLC, and SolarWorld Industries Sachsen GmbH, are both companies involved in the manufacture of components for solar power products. Prior to the lawsuit, the two...
-
On July 31, 2012, Fraiser Company had a cash balance per books of $6,140. The statement from Nasshota State Bank on that date showed a balance of $7,690.80. A comparison of the bank statement with...
-
As a X Engineer in a design consultancy company, you have purchased a high specification computing system. The computing system has a cost basis of USD 4 0 0 0 and a ten - year depreciable life. The...
-
The management of Madeira Computing is considering the introduction of a wearable electronic device with the functionality of a laptop computer and phone. The fixed cost to launch this new product is...
-
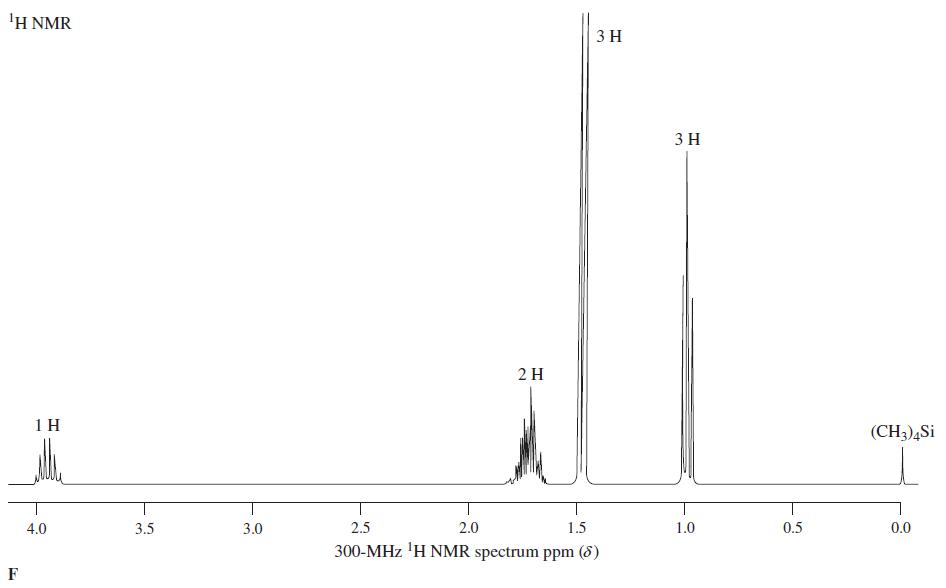
Reaction of the compound corresponding to spectrum E with SOCl 2 produces a chloroalkane, C 4 H 7 Cl, whose NMR spectrum is almost identical with spectrum E, except that the broad signal at = 1.5...
-
Give the structure of an alkene that will give the following carbonyl compounds upon ozonolysis followed by reduction with (CH 3 ) 2 S. (a) CH 3 CHO only (b) CH 3 CHO and CH 3 CH 2 CHO (c) (CH 3 ) 2...
-
Find three examples of lessons from the front line that are evident in the Nestl case. How could these issues be overcome?
-
Explain how to price a bond issuance. If Mark Industries issues $2,000,000 of 10 year bonds on Jan. 1st, 2023. They have a coupon or stated rate of 6% paying semi-annual interest on 6-30 and 12-31....
-
The mass of a planet is 6.5x10^(24) kilograms. Its volume is 1.7x10^(12) cubic kilometers. Find the density of the planet ((mass )/(volume )) in units of grams per cubic centimeter.
-
OKR (Objectives and Key Results) is a goal-setting framework used by organizations to define and track progress towards specific, and measurable outcomes and many organizations use OKR as their...
-
Shakila owns a chain of successful grocery stores that sell lottery tickets. Kaitlin bought a ticket this morning. Kaitlin won $1,000,000 in the lottery. $50,000 per year for 20 years. Kaitlin has a...
-
Information for Computing Gross Margin Description Amount Explanation Sales 100,000 Cost of goods sold 38,000 21,000 fixed + 17,000 variable Operating expenses 30,000 18,000 fixed + 12,000 variable...
-
Describe the process the IRS uses to collect the tax that is found to be due after an audit is completed. Assume that the IRS findings are not appealed but that the taxpayer does not pay the amount...
-
Read the Forecasting Supply Chain Demand Starbucks Corporation case in your text Operations and Supply Chain Management on pages 484-485, then address the four questions associated with the...
-
A laser used to read CDs emits red light of wavelength 700 nm. How many photons does it emit each second if its power is? (a) 0.10 W, (b) LOW?
-
The work function for metallic cesium is 2.14 eV. Calculate the kinetic energy and the speed of the electrons ejected by light of wavelength (a) 700 nm, (b) 300 nm.
-
Calculate the size of the quantum involved in the excitation of (a) An electronic oscillation of period 2.50 fs, (b) A molecular vibration of period 2.21 fs, (c) A balance wheel of period 1.0 ms....
-
On January 1, BBA borrows $192,000 from Citizen Bank. The loan is due in one year along with APR of 8% interest. The company is preparing its quarterly report for March 31. Explain that what best...
-
Bond X is a premium bond making semiannual payments. The bond has a coupon rate of 8.8 percent, a YTM of 6.8 percent, and has 13 years to maturity. Bond Y is a discount bond making semiannual...
-
In order to explain the US defense budget, you are using the data from 1962 to 1981 with the following variables (all measured in billions USD) and estimate the corresponding model (Model 1):(Use...

Study smarter with the SolutionInn App


