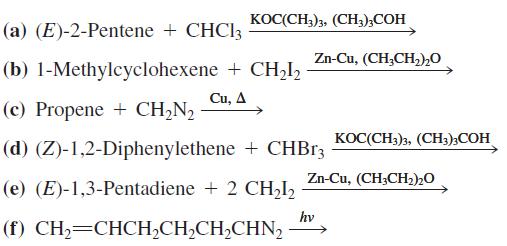
Write the expected products of each of the following reactions. KOC(CH3)3, (CH,);COH (a) (E)-2-Pentene + CHCI3 Zn-Cu,
Question:
Write the expected products of each of the following reactions.
Transcribed Image Text:
KOC(CH3)3, (CH,);COH (a) (E)-2-Pentene + CHCI3 Zn-Cu, (CH,CH,)2O (b) 1-Methylcyclohexene + CH,I, Си, Д (c) Propene + CH,N, KOC(CH3)3, (CH;);COH (d) (Z)-1,2-Diphenylethene + CHB13 Zn-Cu, (CH;CH2)20 (e) (E)-1,3-Pentadiene + 2 CH2I2 hv (f) CH,=CHCH,CH,CH,CHN,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
a We get ccl 2 as a carbene which got inserted into the duoble bond of the alkene b This is ...View the full answer

Answered By

Unik arora
right now i'm in final semester of my masters in chemistry . my strong space is organic and physical chemistry.
till now i don't have any tutoring experience but my main focus is always on build fundamental of subject .so my teaching / studying methodology is based on fundamental . i cleared a bug of easy exams like IIT JAM ,CSIR-NET,GATE ,JEE MAINS
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Suggest a reasonable mechanism for each of the following reactions:
-
Write structural formulas for the major organic products from each of the following reactions. (a) (b) (c) (d) (e) (f) (g) (h) O (excess), H SO (cat.) OH 0 H2O, H,so, (cat.) (1) Mg (2) CO2 (3) H2O Br...
-
Write structural formulas for the major organic products from each of the following reactions. (a) (b) (c) (d) (e) CI CH CH.SH NH2 (excess Cl OH H SO, (cat.) HO AlCl3
-
Suggest two reasons why the adjustments proposed by independent auditors more often than not call for reducing recorded earnings.
-
Build a quality management plan for the other two R & S Amusements Services selected projects customer contract management or collection management.
-
Are most businesses in the United States corporations? If not, what are most businesses?
-
Extreme Machine manufactures machines and parts for various industries; they have an office in Youngstown, Ohio. Avery Dennison manufactures and sells labels from a plant in Mentor, Ohio. They use...
-
Game, LLP, is evaluating the performance of three divisions: Rock, Scissors, and Paper. Using the data that follow, compute the economic value added by each division, and comment on each...
-
A manufacturing company reports the following information. Raw materials inventory, ending Raw materials used Current Year $ 169,500 2,160,000 1 Year Ago 2 Years Ago $ 190,500 2,522,000 $ 197,500...
-
1. Why do you think InnovationCo used the specific standardization and knowledge networking approach in IHRM? What other options did it have? 2. What are the advantages and disadvantages associated...
-
Propose a mechanism for the peroxide-initiated reaction of CH 3 SH with 1-hexene.
-
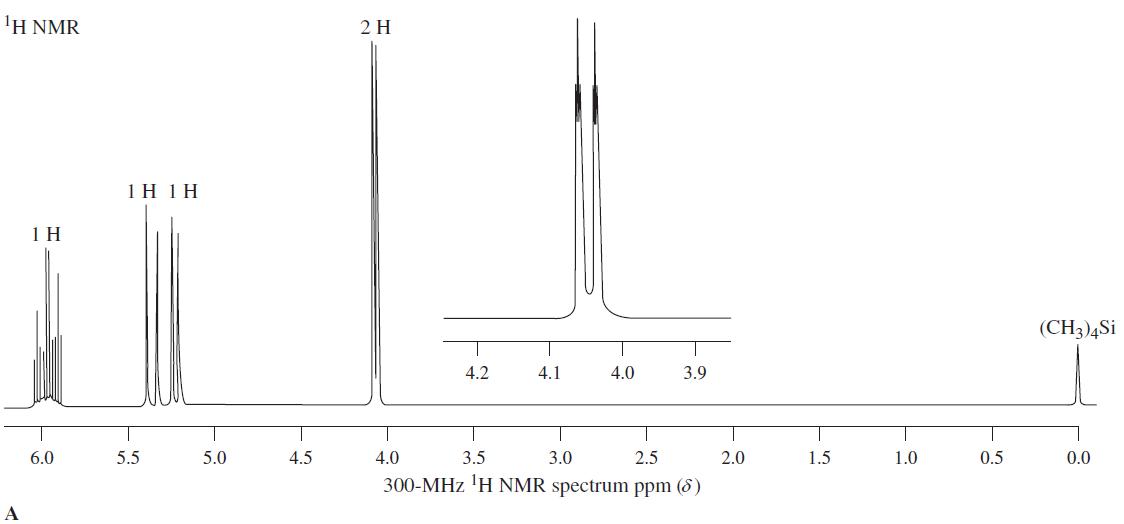
1 H NMR spectrum A corresponds to a molecule with the formula C 3 H 5 Cl. The compound shows significant IR bands at 730 (see Problem 53 of Chapter 11), 930, 980, 1630, and 3090 cm -1 . (a) Deduce...
-
Find the observed, normal, and standard times for the following jobs shown on the website. Assume standard performance and a 10 percent allowance. a) Stamping extrusions b) Stamping end couplings c)...
-
What is the purpose of project risk management? Explain how this is to be undertaken and give some examples of risks faced in a project. Assess the implications when effective risk management...
-
As part of its public sector modernization effort, a North African government has put a lot of effort and allocated significant budgets since 1997 to modernize one of its main public sector...
-
what are Risk Analysis and Mitigation; Example: Unavailability of suitable lease space may delay project deliverables. Possible mitigation strategy: Work with a realtor to identify and screen lease...
-
We learned about different sources of power. Examples include: Coercive power Relational power Guilt-based power Expert power Information power Avoiding power a. Define each of these six sources of...
-
What kind of financial assistance does a consumer receive who qualifies for Low Income Subsidy? Lower or no monthly plan premiums and lower or no copayments A refund based on a predetermined...
-
Elijah pays for his widowed mother's heart bypass surgery. He pays the surgical team directly and gives his mother the money for the hospital bill. Comment on Elijah's gift tax consequences.
-
[a] Two foam blocks, each with a charge of 19 micro coulombs (1 C = 10-6 C), are both held in place 19 cm apart in the east-west direction. A foam ball with a charge 49 C is placed 55 cm north of the...
-
Express the equilibrium constant of a gas-phase reaction A + 3 B ~ 2 C in terms of the equilibrium value of the extent of reaction, ~, given that initially A and B were present in stoichiometric...
-
Show that, if the ionic strength of a solution of the sparingly soluble salt MX and the freely soluble salt NX is dominated by the concentration C of the latter, and if it is valid to use the...
-
To get a sense of the effect of cellular conditions on the ability of ATP to drive biochemical processes, compare the standard Gibbs energy of hydrolysis of ATP to ADP with the reaction Gibbs energy...
-
The Singapore site has also been subnetted to allow for more branches in future. There is already one LAN at the Singapore site => 192.168.1.64/27 This LAN is the one that includes the two servers....
-
1. Looking at your work or professional experience, identify two scenarios (one for individual performance and one for organisational performance) that could have been improved through collaboration....
-
1. Give your analysis on how ITIL helps the IT businesses? 2. Discuss the major focus of ITIL? 3.Describe the benefit of ITIL change management? 4. Identify the challenges of ITIL change management?...

Study smarter with the SolutionInn App


