Compute the partition function per particle? for helium gas at NTP confined in a cube of side
Question:
Compute the partition function per particle?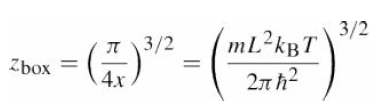
for helium gas at NTP confined in a cube of side 1 cm. What quantum state has the highest probability of being occupied? What is the probability that a given helium atom is in this state? Given the number of atoms in the box, what is the expected average occupation number of this state?
Transcribed Image Text:
3/2 I 3/2 mL2kBT Zbox %3D %3D 4.x 2n h?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
The ground state has with l m n 1 has the largest p...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Physics questions
-
It was asserted that in order for entropy to be an extensive quantity, the classical partition function of a system of indistinguishable particles must be divided by N!. Consider a box evenly...
-
Evaluate the translational partition function for Ar confined to a volume of 1000 cm 3 at 298 K. At what temperature will the translational partition function of Ne be identical to that of Ar at 298...
-
Determine the total molecular partition function for I 2 , confined to a volume of 1000 cm 3 at 298 K. Other information you will find useful: B = 0.0374 cm 1 , v = 208 cm 1 , and the ground...
-
Jogger 1 is travelling east at 6 . 5 m / s and has a mass of 8 2 kg . Jogger 2 is travelling north at 5 . 8 m / s and has a mass of 5 4 . 5 kg . One of the joggers has their head down and doesnt see...
-
The drawing shows two identical stones attached to cords that are being whirled on a tabletop at the same speed. The radius of the larger circle is twice that of the smaller circle. How is the...
-
Grand Canyon Manufacturing Inc. produces and sells a product with a price of $100 per unit. The following cost data have been prepared for its estimated upper and lower limits of activity: Required:...
-
Arthur Shore practiced accounting with a partnership for five years. Recently he opened his own accounting firm, which he operates as a professional corporation. The name of the new entity is Arthur...
-
The comparative balance sheet of Whitman Co. at December 31, 2016 and 2015, is as follows: The noncurrent asset, noncurrent liability, and stockholders' equity accounts for 2016 are as follows: Dec....
-
How does international credit management differ from credit management in a single country? Company would any type of international credit management to help their company succeed in other countries....
-
The file P02_35.xlsx contains (fictional) data from a survey of 500 randomly selected households. a. Indicate the type of data for each of the variables included in the survey. b. For each of the...
-
Use the Sackur?Tetrode equation? ? to show that (?S/?U)|V = 1/T for an ideal monatomic gas. Likewise show that (?S/?H)|p = 1/T, verifying eq and the assertion of footnote 6. Sideal = NkB + In V + 5...
-
Real crystals contain impurities, which lead to nonzero entropy at T = 0. Consider a crystal consisting of N atoms. The crystal is primarily composed of element A but contains M N atoms of an...
-
Two record companies, A and B, produce classical music recordings. Label A is a budget label, and 5% of A's new compact discs exhibit significant degrees of warpage. Label B is manufactured under...
-
An irreversible process takes place by which the entropy of the universe increases by 1.66 J/K. If the temperature of the environment in which the process occurred is 225 K, how much energy (in J)...
-
Give statistics for each travel agency for the years 2 0 1 0 through 2 0 2 2 , including the profit and loss for each ( note the value with ' - ' for loss ) . Additionally, find out how many people...
-
Plato Ltd Trial Balance as at 31st October 2009 was balanced by the inclusion of the following debit balance of $5,500 in the Suspense Account Subsequent investigation revealed the following errors 9...
-
Refer to the original data. Assume that the company sold 42,000 units last year. The sales manager is convinced that a 2% reduction in the selling price, combined with a $152,000 increase in...
-
Radar was very important during World War II. It used electromagnetic waves to de- tect, for example, the location of planes relative to a ground location. Discuss whether such radar set-ups would be...
-
Describe the type of fees that you would pay to purchase a load fund. What annual fees would you typically pay for your mutual fund investment?
-
Decades after the event, Johnson & Johnson (J&J), the 130-year-old American multinational, is still praised for swiftly The company indicated that its response was based on the expectations set forth...
-
Suppose the solid cylinder in the apparatus described in Example 9.8 (Section 9.4) is replaced by a thin-walled, hollow cylinder with the same mass M and radius R. The cylinder is attached to the...
-
A frictionless pulley has the shape of a uniform solid disk of mass 2.50 kg and radius 20.0 cm. A 1.50-kg stone is attached to a very light wire that is wrapped around the rim of the pulley (Fig....
-
A bucket of mass m is tied to a massless cable that is wrapped around the outer rim of a frictionless uniform pulley of radius R, similar to the system shown in Fig. E9.47. In terms of the stated...
-
18. During your years as a wise college freshman you racked up $3,478 in credit card debt. The card's APR is 17.99%, and you plan to pay off the card with reasonable monthly payments of $150 per...
-
is discussion activity features a dream vacation of your choice and associated annuity calculations. Tell us about your dream vacation. Do some quick research to find out how much this dream vacation...
-
Based on the brief transcript/ scenario below and the cuts that are being proposed, what would be the best solution in creating an operating budget for the department? Do you take a 'salami slice'...

Study smarter with the SolutionInn App


