A 0.505-in.-diameter metal bar with a 2-in. gage length l0 is subjected to a tensile test. The
Question:
A 0.505-in.-diameter metal bar with a 2-in. gage length l0 is subjected to a tensile test. The following measurements are made in the plastic region:
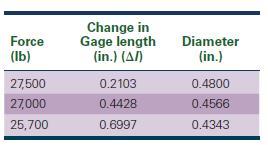
Determine the strain-hardening exponent for the metal. Is the metal most likely to be FCC, BCC, or HCP? Explain.
Transcribed Image Text:
Force (lb) 27,500 27,000 25,700 Change in Gage length (in.) (A/) 0.2103 0.4428 0.6997 Diameter (in.) 0.4800 0.4566 0.4343
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
My explanation is that the metal is most likely BCC Due to the linear relationship t...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
The board is subjected to a tensile force of 200 lb. Determine the average normal and average shear stress in the wood fibers, which are oriented along plane aa at 20° with the axis of the board....
-
The yoke-and-rod connection is subjected to a tensile force of 5 kN. Determine the average normal stress in each rod and the average shear stress in the pin A between the members. 40 mm 5 kN. 30 mm...
-
A piece of material is subjected to a tensile stress of 50 N/mm2 in one direction and compressive stress of 30N/mm2 in a direction perpendicular to the previous one. Find the stress on a plane the...
-
Multiple Select Question Select all that apply Identify what might happen if a particular department were eliminated. (Check all that apply.) Multiple select question. Many of the indirect expenses...
-
Distinguish between the obligation placed on a bailee for value and that imposed on a common carrier or innkeeper.
-
Use a graphing utility to solve equation. Express the solution(s) rounded to two decimal places. 19x + 8 cos x = 2
-
Derive an expression for the heat loss conductance \(U_{c}\) for a flat-plate collector in which convection and conductance are completely eliminated in the air layers by use of a hard vacuum.
-
Hawk Homes, Inc., makes one type of birdhouse that it sells for $30 each. Its variable cost is $15 per house, and its fixed costs total $13,840 per year. Hawk currently has the capacity to produce up...
-
Selected data (in thousands) from the statement of income of Foress Inc. are shown below. Using vertical analysis, calculate the percentage of the base amount for each year. (Round answers to 1...
-
Ted James, the accountant for Andersen Company, must complete Form 941 for the first quarter of the current year. Ted gathered the needed data as presented in Problem 8A-1 . Suddenly called away to...
-
A 1.33-cm-diameter metal bar with a 3-cm gage length (l 0 ) is subjected to a tensile test. The following measurements are made in the plastic region: Determine the strain-hardening coefficient for...
-
An axially loaded compression member is cold formed from an AISI 10 -5 steel plate. A cross-section of the member is shown in Figure K8-1. The metal at the corners of the member was strengthened by...
-
Use the PPF framework to explain something in your everyday life that was not mentioned in the chapter.
-
Solve: log2 (x-2)-log2 (6x-5)= 0.
-
Solve the equation for b. 2b2+21b+54=0
-
The Budget component must be constructed fora Marketing Strategy for the Model X (Tesla automobile). However,I'm having trouble coming up with a break-even analysis. Please provide an analisys that...
-
A bank charges Strong Co. an interest rate of 5.75% compounded quarterly on its line of credit. If 0.75% was added to the effective interest rate, calculate the new interest rate compounded...
-
Light emitting diodes (LED) light bulbs have become required in recent years, but do they make financial sense? Suppose a typical 60-watt incandescent light bulb costs $.48 and lasts 1,000 hours. A...
-
Why do both thymine and uracil specify the incorporation of adenine?
-
The graph of the sequence whose general term is an = n - 1 is which of the following? [8.1] A. B. TITTT 3-2-1 23.45 2.3.4
-
For the reaction K = 2.4 10 -3 at a given temperature. At equilibrium in a 2.0-L container it is found that [H 2 O(g)] = 1.1 10 -1 M and [H 2 (g)] = 1.9 10 -2 M. Calculate the moles of O 2 (g)...
-
Ethyl acetate is synthesized in a nonreacting solvent (not water) according to the following reaction: For the following mixtures (ad), will the concentration of H 2 O increase, decrease, or remain...
-
A 1.00-L flask was filled with 2.00 moles of gaseous SO 2 and 2.00 moles of gaseous NO 2 and heated. After equilibrium was reached, it was found that 1.30 moles of gaseous NO was present. Assume that...
-
Assume you are a trader with JP Morgan. From the quote screen on your computer terminal, you notice that Bank A is quoting 0.8354/$1.00 and Bank Bis offering SF1.0913/$1.00. You learn that Bank Cis...
-
Leven Clinic uses client-visits as its measure of activity. During September, the clinic budgeted for 3,000 client-visits, but its actual level of activity was 3,050 client-visits. The clinic has...
-
If 2.25 grams of calcium hydroxide is dissolved in 154.56 ml of water, a. What's the Molarity (M), if the volume of solution is 500.00 ml? b. What's the Molarity (M) of hydroxide ions, if the volume...

Study smarter with the SolutionInn App


