A steel microstructure contains 92% martensite and 8% Fe 3 C; the composition of the martensite is
Question:
A steel microstructure contains 92% martensite and 8% Fe3C; the composition of the martensite is 1.10% C. Using Figure 12-35, determine
(a) The temperature from which the steel was quenched and
(b) The carbon content of the steel.
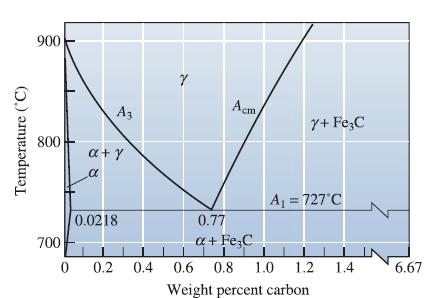
Transcribed Image Text:
Temperature (°C) 900 800 700 0 A3 a+y α 0.0218 0.2 0.4 Y I Acm Y+ Fe C A₁ = 727°C 0.77 a + Fe C Tī 0.6 0.8 1.0 1.2 1.4 Weight percent carbon 6.67
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
a The temperature 1250C so that the martensite is in ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
A steel microstructure contains 75% marten site and 25% ferrite; the composition of the marten site is 0.6% C. Using Figure 12-32, determine (a) The temperature from which the steel was quenched and...
-
The composition of timothy hay is 11.3% protein, 1.6% fat and 41% crude fiber (as-fed basis). How many pounds of protein, fat, and crude fiber are present in a ton of hay?
-
The composition of moist air is given on a molar basis to be 78 percent N2, 20 percent O2, and 2 percent water vapor. Determine the mass fractions of the constituents of air.
-
You are in the market for a new car and have narrowed your search down to two types: Car X costs $36,300, will last for four years, and will be worth $3,630 at the end of its useful life. It will...
-
Without looking at the text, derive a nations offer curve from its production frontier, its indifference map, and two relative commodity prices at which trade could take place (i.e., sketch a figure...
-
Geraldine Wolfe is a supervisor at Fantastigifts. She has an annual salary of $45,000, paid biweekly, and a garnishment for consumer credit of $375. Assuming that her disposable income is 80 percent...
-
Demonstrate how EVA calculations are made.
-
Starkey Companys sales, current assets, and current liabilities (all in thousands of dollars) have been reported as follows over the last five years (Year 5 is the most recent year): Required: 1....
-
What the legal framework for cryptocurrencies in South Africa is. I need legal questions to help me get information to speak about the legal framework of cryptocurrencies in South Africa. I need all...
-
(a) Consider the following time series data. Quarter Year 1 Year 2 Year 3 1 2 4 5 2 4 5 8 3 1 3 4 4 7 9 10 (b) Use a multiple regression model with dummy variables as follows to develop an equation...
-
What step controls the rate of martensitic phase transformations?
-
What is the difference between isothermal and athermal transformations?
-
In Problems 11 22, graph each inequality. x2 x + y2 9
-
A system that uses the Banker's Algorithm has six processes and four types of reusable resources (R1, R2, R3 and R4). The current allocation, maximum needs (claims) and available resources are as...
-
How does the use of artificial intelligence (AI) and machine learning (ML) in the creation of intellectual property impact traditional notions of copyright and patent law?
-
How can faculty ascertain whether the curriculum framework is providing adequate structure to support decisions faculty make regarding curriculum development and implementation?2. What are effective...
-
What leaders or leader do you think the instWhat leaders or leader do you think the instructor is talking about?ructor is talking about?
-
What you are really writing about is theperformancewhich you saw. You are writing about the work that the actors have done onstage. You are writing about the technical elements of the production. You...
-
For each of the following reactions, give the elimination products; if the products can exist as stereoisomers, indicate which stereoisomers are obtained. a. (2S.3S-2-chIoro-3-methyIpcntane + high...
-
Q1) What is the a3 Value Q2) What is the a7 Value Q3) What is the a4 Value Q4) What is the b3 Value Q5) What is the b2 Value Q6) What is the sign of 2nd constraint? A pastry chef at a bakery wants to...
-
An automobile assembly plant produces 400 vehicles per day. Make an order-of-magnitude estimate for the weight of the steel needed to make those vehicles. Explain and justify the reasonableness of...
-
Think of some quantity that you encounter in your day-to-day life for which it would be difficult to obtain a highly accurate numerical value but for which an order-of-magnitude approximation can be...
-
The modulus of elasticity, modulus of rigidity, Poissons ratio, and the unit weight for various materials are shown below. The data is given as Material; Modulus of Elasticity, E (Mpsi & GPa);...
-
Given the matrix A -3 0 -14 3 4 6 9115 7701 a) Determine all solutions of the homogeneous system Ax = 0. b) Determine if the columns of A span R.
-
Summarize each data source and include them for substance abuse and alcohol in the military. Analyze each data source for substance abuse and alcohol in the military for trustworthiness and accuracy....
-
On January 1, 2023, Holland Corporation paid $7 per share to a group of Zeeland Corporation shareholders to acquire 60,000 shares of Zeeland's outstanding voting stock, representing a 60 percent...

Study smarter with the SolutionInn App


