Common commercial acids and bases are aqueous solutions with the following properties: Calculate the molarity, molality, and
Question:
Common commercial acids and bases are aqueous solutions with the following properties:
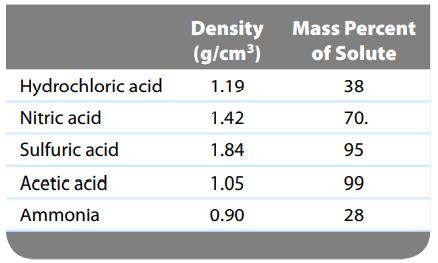
Calculate the molarity, molality, and mole fraction of each of the preceding reagents.
Transcribed Image Text:
Hydrochloric acid Nitric acid Sulfuric acid Acetic acid Ammonia Density (g/cm³) 1.19 1.42 1.84 1.05 0.90 Mass Percent of Solute 38 70. 95 99 28
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
To calculate the molarity molality and mole fraction of each reagent we need to use the given inform...View the full answer

Answered By

Dulal Roy
As a tutor, I have gained extensive hands-on experience working with students one-on-one and in small group settings. I have developed the ability to effectively assess my students' strengths and weaknesses, and to customize my teaching approach to meet their individual needs.
I am proficient at breaking down complex concepts into simpler, more digestible pieces, and at using a variety of teaching methods (such as visual aids, examples, and interactive exercises) to engage my students and help them understand and retain the material.
I have also gained a lot of experience in providing feedback and guidance to my students, helping them to develop their problem-solving skills and to become more independent learners. Overall, my hands-on experience as a tutor has given me a deep understanding of how to effectively support and encourage students in their learning journey.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Calculate the mass fraction and mole fraction of each component and the gas constant of the mixture for each of the following mixtures: a) 2 lbm N2, 3 lbm O2, 5 lbm CO2 b) 3 lbmol CH4, 4 lbmol N2, 3...
-
Calculate the ratio [NH 3 ]/[NH 4 + ] in ammonia/ammonium chloride buffered solutions with the following pH values: a. pH = 9.00 b. pH = 8.80 c. pH = 10.00 d. pH = 9.60
-
Calculate the molality of each of the following aqueous solutions: (a) 2.50 M NaCl solution (density of solution = 1.08 g/mL), (b) 48.2 percent by mass KBr solution.
-
Correction, overproduction, inventory and motion are all examples of: a. Waste b. 5 S target areas c. Noise d. Value-added activities
-
List the criteria marketers use to determine whether a segment may be a good candidate for targeting.
-
In the figure is a proposed shaft design to be used for the input shaft a in Prob. 77. A ball bearing is planned for the left bearing, and a cylindrical roller bearing for the right. (a) Determine...
-
Five hundred null hypotheses are tested at the = 0.05 level, and 40 of them are rejected. Assume the tests are independent, a. Assuming that all 500 null hypotheses are true, what is the probability...
-
Clown Around, Inc., provides party entertainment for children of all ages. The following is the company's trial balance dated February 1, current year. Clown Around engaged in the following...
-
Conde Inc. has projected sales to be: February, $20,000; March, $18,000; April, $16,000; May, $20,000; and June, $22,000. Conde has 30% cash sales and 70% sales on account. Accounts are collected 40%...
-
A demand loan of $45,000 on November 05, 2020 at 5% p.a. simple interest was settled by a payment of $700 on March 15, 2021 and the outstanding balance on June 15, 2021. Using the declining balance...
-
What is ion pairing?
-
Explain the terms isotonic solution, crenation, and hemolysis.
-
Define what is meant by a stakeholder and name five stakeholders and describe their expectations from the organization.
-
Explain how government intervention in markets might not improve market outcomes.
-
Is it possible for a firm or firms operating in a natural monopoly to be subject to contestability? Explain.
-
How would an extension of property rights help reduce the instances of market failure?
-
Use an example to explain the idea of a positional externality. Example Positional Externalities Positional goods have the characteristic that the utility from consumption of a good is dependent on...
-
In a perfectly contestable market, inefficiency does not exist. Explain why.
-
Explain how the following events would likely affect the relevant interest rate. a. A major bond-rating agency has improved the risk rating of a developing nation. b. The government has passed...
-
XYZ Inc. a calendar year, accrual basis corporation, had the following items during 2021: Gross revenue from operations Cost of goods sold $420,000 ($180,000) $9,000 LT capital gain .LT capital...
-
A differential dz = f (x, y) dx + g(x, y) dy is exact if the integral f (x, y) dx + g(x, y) dy is independent of the path. Demonstrate that the differential dz = 2xy dx + x 2 dy is exact by...
-
Make a graph of the densities of a liquid and its gas as a function of the temperature from low temperatures up to the critical point. Explain the behavior at the critical point.
-
Explain why ethene has a higher value for CV ,m at 800. K than CO.
-
The United States, Austrailia, Canada, and the United Kingdom cluster high on individualism but low on power distance on Hofstede\'s cutlutral graph. What does this mean to someone looking to market...
-
Mini-garden project Week one of the mini-garden project involved Sacha and her fellow pre-schoolers planting sunflower seeds into the outdoor garden bed. Each child in the activity has been assigned...
-
A) Translate the following C into RV641 assembly. Be sure to follow the calling (Refer to slide22 of lec6_postZoom.pdf) and comment each line of your assembly. Remember that long long is 64 bits in...

Study smarter with the SolutionInn App


