Complete and balance each acidbase reaction. a. HPO4(aq) + NaOH(aq) Contains three acidic hydrogens b. HSO4(aq)
Question:
Complete and balance each acid–base reaction.
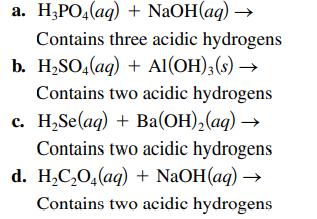
Transcribed Image Text:
a. H₂PO4(aq) + NaOH(aq) → Contains three acidic hydrogens b. H₂SO4(aq) + Al(OH)3(s) → Contains two acidic hydrogens c. H₂Se(aq) + Ba(OH)₂(aq) → Contains two acidic hydrogens d. H₂C₂O4(aq) + NaOH(aq) → Contains two acidic hydrogens
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a H3PO4aq 3NaOHaq Na3PO4aq 3H2Ol b H2SO4aq 2AlOH3s Al2SO43s 6H...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Complete and balance each combustion equation. a. C4H9OH + O2 ( ? b. CH3NO2 + O2 ( ?
-
Complete and balance each combustion equation. a. B2H6 + O2 ( ? (The oxide of boron formed is B2O3.) b. Al2S3 + O2 ( ? (The oxide of sulfur formed is SO2.) c. Al2S3 + O2 ( ? (The oxide of sulfur...
-
Complete and balance each of the following molecular equations, including phase labels, if a reaction occurs. Then write the net ionic equation. If no reaction occurs, write NR after the arrow. a....
-
1. Prepare a schedule of cost of goods manufactured for Denim Bones for the year ended December 31, 2024. 2. Prepare an income statement for Denim Bones for the year ended December 31, 2024. 3. How...
-
Martha Lou owns 100 shares of Blain Corporation common stock. She purchased the stock on July 25, 1986, for $4,000. On May 2 of the current year, she receives a nontaxable distribution of 100 stock...
-
Consider the differential equation \[ \ddot{x}+2 \zeta \omega_{n} \dot{x}+\omega_{n}^{2} x=0 \] Define in words and in terms of system parameters \(m\), \(c\), and \(k\) for (a) \(\omega_{n}\) and...
-
The numbers of working hours of a sample of 30 employees in a month Use technology to draw a box-and-whisker plot that represents the data set. 160 182 195 196 174 196 210 199 187 164 131 211 238 132...
-
Lawrence Corporation sells two ceiling fans, Deluxe and Basic. Current sales total 60,000 units, consisting of 39.000 Deluxe units and 21,000 Basic units. Selling price and variable cost information...
-
ABC Boat Company is interested in replacing a moulding machine with a new improved model. The old machine has a salvage value of $ 2 0 , 0 0 0 now and a predicted salvage value of $ 4 , 0 0 0 in six...
-
A vehicle has the following system parameters: total mass, m (kg): 2037 wheel base, L (m): 2.794 Estimate the possible maximum speed of the vehicle on level ground and on a grade of 25% as determined...
-
You made 100.0 mL of a lead(II) nitrate solution for lab but forgot to cap it. The next lab session you noticed that there was only 80.0 mL left (the rest had evaporated). In addition, you forgot the...
-
In the spectroscopic analysis of many substances, a series of standard solutions of known concentration are measured to generate a calibration curve. How would you prepare standard solutions...
-
Shalie Manufacturing makes several thousand different metal products, including building materials, tools, and furniture parts. The manager of the furniture parts division has proposed that his...
-
Based on the AHf data given, which compound is the most stable? HS(g), -20.6 kJ/mol NH4(g), +94.5 kJ/mol O PH3(g), +5.4 kJ/mol NH3(g), -46.4 kJ/mol NO4(g), +9.7 kJ/mol
-
16 Methanol is often used as an "antifreeze" for windshield washer fluid. How many moles of methanol would have to be added to 1000g of water to lower the freezing point of water to -20C? The...
-
What will be the current of electricity in 20m when 0.012mole of oxygen molecule during the electrolysis of dilute CUS04 solution [if =96500]
-
A chemical reaction is run in which 188 Joules of heat are generated and 264 Joules of work is done on the system. Calculate the change in the internal energy of the chemical system.
-
A student is trying to identify the composition of a pure metal block by determining its density. The student records the mass of the sample to be 2 5 . 0 g . The rectangular block is 3 . 0 0 cm \...
-
Briefly describe three ways that antibody diversity is produced.
-
Ashlee, Hiroki, Kate, and Albee LLC each own a 25 percent interest in Tally Industries LLC, which generates annual gross receipts of over $10 million. Ashlee, Hiroki, and Kate manage the business,...
-
Which of the following molecules may show a pure rotational Raman spectrum? (a) CH2Cl2 (b) CH3CH3, (c) SP6, (d) N2O?
-
An object of mass 2.0 g suspended from the end of a spring has a vibrational frequency of 3.0 Hz. Calculate the force constant of the spring.
-
Calculate the percentage difference in the fundamental vibration wave number of IH35Cl and 2H37CIon the assumption that their force constants are the same.
-
Determine "Tyson Food "Company's firmographics, activities, and objectives. Then relate these differences to differences in the organizational cultures of the organizations. or provide detail of any...
-
Wimble Ltd had $400 million of debt outstanding at an interest rate of 9% and $600 million of equity (market value) outstanding. Wimble is subject to a 30% corporate tax rate. What is the amount of...
-
explain and comments thies pints Has existed for a long time One of the original fast-food establishments High standing Commonly recognized for its root beer. With high-quality cuisine Markets to...

Study smarter with the SolutionInn App


