Complete the table for each of the following solutions: 0.0070 M HNO3 3.0 M KOH [H+] PH
Question:
Complete the table for each of the following solutions:
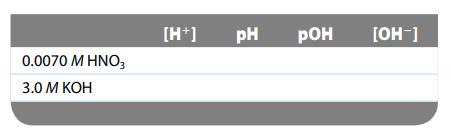
Transcribed Image Text:
0.0070 M HNO3 3.0 M KOH [H+] PH РОН [OH-]
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
To fill in the table use the equations that relate pH pOH and the concen...View the full answer

Answered By

Saleem Abbas
Have worked in academic writing for an a years as my part-time job.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Complete the following ANOVA table for each of the two dot plots: For Information: Refer to Exercises 9.17 and 9.18. Source df SS MS Treatments Error Total
-
Complete the chart for each of the following independent distributions. Assume that all distributions are nonliquidating and pro rata to the partners, that no contributed property was distributed,...
-
Complete the table for the time t (in years) necessary for P dollars to triple when it is invested at an interest rate r compounded (a) Continuously (b) Annually. 6% 2% 4% 8% 10% 12%
-
In a world so divided in so many ways, sustainable economic development on a global scale will amount to nothing but a figment of human imagination or, to put it mildly, just wishful thinking. Do you...
-
Define the law of large numbers. What are its implications for an insurance system?
-
In 2008, Gogo became the first company to offer Wi-Fi service on commercial aircraft. It provides the service primarily through ground-based cellular towers. Many air travelers find the $30 price...
-
Derive the stiffness and mass matrices of the planar frame element (general beam element) shown in Fig. 12.16 in the global \(X Y\)-coordinate system. Y U U3 U Joint 1 Joint 2 U6 Us UA FIGURE 12.16 A...
-
The L. Young & Sons Manufacturing Company produces two products, which have the following profit and resource requirement characteristics: Last months production schedule used 350 hours of labor in...
-
Write a thank you response to the following text "Thanks for this clear and concise post on Municipal Bonds. I believe that even though municipal bonds offer tax advantages to higher income...
-
Sunshine Sushi, a Japanese restaurant, has the following adjusted trial balance with accounts listed in alphabetical order. Using the template provided, prepare a classified balance sheet for the...
-
Consider 0.10 M solutions of the following compounds: AlCl 3 , NaCN, KOH, CsClO 4 , and NaF. Place these solutions in order of increasing pH.
-
Calculate the pH of a 1.0 10 -7 -M solution of NaOH in water.
-
Suppose that on October 24, 2016, a company sells one April 2017 live-cattle futures contract. It closes out its position on January 21, 2017. The futures price (per pound) is 151.20 cents when it...
-
Given that Rahn and Deepa want to equally own the new property, claim taxation benefits and maintain ownership if one party passes away, how should they register their ownership interest on the...
-
Cash flows are an important indicator of a company's financial health. What is cash flow and what would you do if your company continued to run negative cash flow every quarter? How would you fix the...
-
Briefly discuss the Quantitative and Qualitative methods for forecasting.
-
Murphy, who has retired for several years, is considering investing in two stocks: A and B. The returns and other related estimates for the two stocks under different states of the world in the...
-
(2 points) What is today's value of firm F (A) if firm F's leverage is L=1? Hint: 1) Compute the cash-flows to the shareholders of a comparable (exact same features as F except the capital structure)...
-
Faraday flashlight uses electromagnetic induction to produce energy when shaken. A magnet in the handle is free to slide back and forth through a loop with 50 000 turns. The energy is stored in a...
-
In a nonmagnetic medium, E = 50 cos (10 9 t 8x) a y + 40 sin (10 9 t 8x) a z V/m find the dielectric constant r and the corresponding H.
-
Which of the following compounds cannot be prepared by an acetoacetic ester synthesis? Explain. (a) Phenyl acetone (b) Acetophenone (c) 3, 3-Dimethyl-2-butanone
-
How would you prepare the following compound using an acetoacetic estersynthesis?
-
How might you prepare the following compounds using an alkylation reaction as the keystep? (b) -H CH-CH (a) CH2CH=CH2 (c) CHCH,CH2CHCEN C (d) (f) (e) CH - CH CHCHCHCH CH-CH - Hg, CH(CH3)2
-
Harwood Company uses a job-order costing system that applies overhead cost to jobs on the basis of machine-hours. The company's predetermined overhead rate of $2.60 per machine-hour was based on a...
-
Dahlia Corporation has a current accounts receivable balance of $447,016. Credit sales for the year just ended were $4,950,605. a. What is the receivables turnover? Note: Do not round Intermediate...
-
Crenshaw Inc. is considering a change to activity-based product costing. The company produces two products, cell phones and tablet PCs, in a single production department. The production department is...

Study smarter with the SolutionInn App


