Consider the following exothermic reaction at equilibrium: Predict how the following changes affect the number of moles
Question:
Consider the following exothermic reaction at equilibrium:
![]()
Predict how the following changes affect the number of moles of each component of the system after equilibrium is reestablished by completing the table below. Complete the table with the terms increase, decrease, or no change.
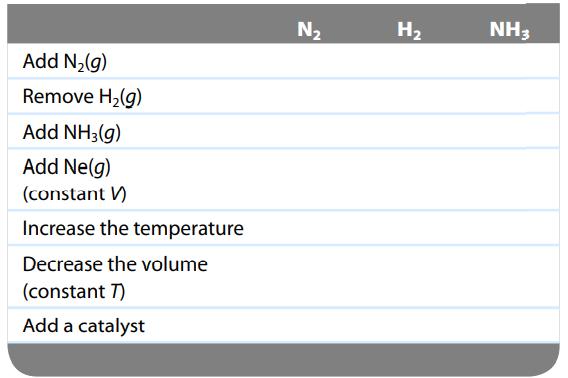
Transcribed Image Text:
)=2NH3(g) N₂(g) + 3H₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
ANSWER Changes N2 H2 NH3 Add N2g Increase Decrease Increase Remove H2g Decrease Decrease Increase Add NH3g Increase Increase Decrease Add Neg No chang...View the full answer

Answered By

Churchil Mino
I have been a tutor for 2 years and have experience working with students of all ages and abilities. I am comfortable working with students one-on-one or in small groups, and am able to adapt my teaching style to meet the needs of each individual. I am patient and supportive, and my goal is to help my students succeed.
I have a strong background in math and science, and have tutored students in these subjects at all levels, from elementary school to college. I have also helped students prepare for standardized tests such as the SAT and ACT. In addition to academic tutoring, I have also worked as a swim coach and a camp counselor, and have experience working with children with special needs.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Complete the following table by entering either increase or decrease in each cell: Item Debit Credit venues Expenses
-
Consider the following reaction: (a) Is this reaction exothermic or endothermic? (b) Calculate the amount of heat transferred when 3.55 g of Mg(s) reacts at constant pressure. (c) How many grams of...
-
Consider the following equilibrium systems: Predict the change in the equilibrium constant Kc that would occur in each case if the temperature of the reacting system were raised. (a) A2B (b) A BC (c)...
-
A trial balance was extracted from the books of V Baker, and it was found that the debit side exceeded the credit side by 40. This amount was entered in the suspense account. The following errors...
-
Explain the steps in distribution planning.
-
Identify the conic that each polar equation represents. Also, give the position of the directrix. 4 2 - 3 sin 0
-
Leicht Transfer & Storage provides warehousing services and often purchases pallets from Pallet Central. The companies followed a standard practice for documenting these transactions in which Pallet...
-
Assume that you have an after-tax cost of capital of 10 percent. Compute the net present value of each of the five projects listed in the following table. Rank the above projects from 1 (best for...
-
2 Charlie's Crispy Chicken (CCC) operates a fast-food restaurant. When accounting for its first year of business, CCC created several accounts. Account Name Description Balance Accounts Payable...
-
Significant Figures 1. How many significant figures are contained in each of the following numbers? 7809700.897 tons 94700. m 28.2890 cm 347600 L 0.07890 pints 120 atoms Rounding 2. Round each of the...
-
At 5000 K and 1.000 atm, 83.00% of the oxygen molecules in a sample have dissociated to atomic oxygen. At what pressure will 95.0% of the molecules dissociate at this temperature?
-
For the following endothermic reaction at equilibrium: which of the following changes will increase the value of K? a. Increasing the temperature b. Decreasing the temperature c. Removing SO 3 (g)...
-
a. If telecommuting equates to hard real estate cost savings, why dont all companies do it? b. Do you think companies would be as supportive if there werent a real estate cost savings? Why or why...
-
Estimate the Cap Rate using the BAND OF INVESTMENT Method in order to estimate the value of a Rental Property using a Loan to Value Ratio of 75% for a 25 years Mortgage at fixed annual rate of 6%....
-
The only journal entry required for payroll accounting is the one that records the statutory deductions withheld from the employees' pay. Explain
-
An all-equity firm has a beta of 1.03. The firm is evaluating a project that will increase the output of the firm's existing products. The market risk premium is 6.9 percent, and the risk-free rate...
-
what is feasibility? what are the success factors required by a project to be successful? what is project objective? What is the role of a project manager in business case. What is procurement route...
-
Your posts could include any of the following: initiating a question regarding this week's course content, responding to your group member's questions, initiating a discussion by commenting on what...
-
Find the center, the vertices, the foci, and the asymptotes of the hyperbola. Then draw the graph. a. x2 - y2 - 2x - 4y - 4 = 0 b. 4x2 - y2 + 8x - 4y - 4 = 0
-
What is the role of business risk analysis in the audit planning process?
-
Sketch an energy diagram that shows a conformational analysis of 2,2-dimethylpropane. Does the shape of this energy diagram more closely resemble the shape of the energy diagram for ethane or for...
-
What are the relative energy levels of the three staggered conformations of 2,3-dimethylbutane when looking down the C2-C3 bond?
-
Draw the ring flip for each of the following compounds: (a) (b) (c) OH I CI
-
An 88-year-old presents to a primary care office with their daughter for concerns of a skin ulceration on the lateral ankle with hair loss. This has been ongoing for months and has recently worsened....
-
Two years after the first round XMP GmbH receives a second offer form B-Capital. B-Capital offers to invest 12,500,000.00 at a 60,000,000.00 pre-money valuation. What is the founders' post-round...
-
NTP Inc. raises its first venture round of 7,000,000.00 at a pre-money valuation of 14,000,000.00 from A-Capital. Before the round, the company had 100,000.00 shares outstanding, solely distributed...

Study smarter with the SolutionInn App


