Consider the following four structures. a. Which of these compounds would have the same physical properties (melting
Question:
Consider the following four structures.
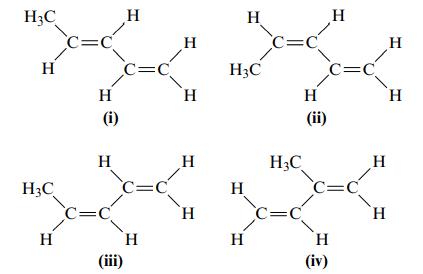
a. Which of these compounds would have the same physical properties (melting point, boiling point, density, and so on)?
b. Which of these compounds are trans isomers?
c. Which of these compounds do not exhibit cis–trans isomerism?
Transcribed Image Text:
H3C H H3C Н c=c H (i) Н c=c н c=c c=c н (iii) н H Н Н H H H3C Н H c=c H H₂C c=c c=c (ii) Н C=C H (iv) H H H H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
a compounds II and III have same physical pr...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Consider the following four structures: a. Which of these compounds have the same physical properties (melting point, boiling point, density, and so on)? b. Which of these compounds are trans...
-
Which of these compounds would you expect to have the highest boiling point? Explain. [Section 24.4] CH3CH CH CH OH CHC=CH HCOCH
-
Consider the structures of cis-decalin and trans-decalin: (a) Which of these compounds would you expect to be more stable? (b) One of these two compounds is incapable of ring flipping. Identify it...
-
Stuart and Belinda, who earn good salaries, want to buy a property they can use to live in and operate a bed and breakfast in their retirement. The only funds they have available is their combined...
-
List some of the forecasting techniques that should be considered when forecasting a trending series. Give examples of situations in which these techniques would be applicable.
-
Combine the reaction entropies calculated in Exercise 3.8b with the reaction enthalpies, and calculate the standard reaction Gibbs energies at 298 K.
-
Master Suites Hotels includes the following selected accounts in its general ledger at December 31,2008 : Prepare the liabilities section of Master Suites' balance sheet at December 31, 2008, to show...
-
An company has sales of $50 million growing at 25% YoY with EBITDA margins at 20%. It secures a JV in Year 3 with additional business of $20 million at EBITDA of 12.5% which linearly scales up to $50...
-
German Seller negotiated with PRC Supermarket for selling 100,000 kilo of living Atlantic Herrings at a price of US78,000. PRC Supermarket showed its strong interest in closing the deal because these...
-
New Wave Shelving's Inventory Manager would like to start using an ABC inventory classification system. The following table shows the annual inventory usage of all the 19 component items that the...
-
Draw the structures for two examples of unsaturated hydrocarbons. What structural feature makes a hydrocarbon unsaturated?
-
Give two examples of saturated hydrocarbons. How many other atoms are bonded to each carbon in a saturated hydrocarbon?
-
For each of the following studies, identify the type of graph (histogram, time series graph, or scatter diagram) that would be the most appropriate. (You can use more than one graph of each type, for...
-
Do you think that all sport organizations should use TQM? Why or why not?
-
Do most sport organizations focus on creating customer value? Use specific examples to defend your position.
-
Do you believe that sport management theory is or should be as precise as physics or chemistry? Explain your answer.
-
For the companies you listed in question 6, identify their methods of going global.
-
Categorize a few sport companies you are familiar with as international or multinational.
-
The citric acid cycle is a series of biological reactions that plays a central role in cell metabolism. The cycle includes dehydration reactions of both malic and citric acids, yielding fumaric and...
-
Suppose that a company has 10.000 outstanding shares in the beginning of the year. On April 1st, the company increases its shares by 6.000. On July 1st, the company increases its shares again, but...
-
The following thermal rearrangement involves two pericyclic reactions in sequence. Identify them, and propose a mechanism to account for the observedresult. 275 "C CD2 - -D H2C CD2
-
Predict the product of the following pericyclic reaction. Is this [5, 5] shift a suprafacial or an antarafacialprocess? [5,5] CH eat
-
Ring-opening of the trans-Cyclobutene isomer shown takes place at much lower temperature than a similar ring-opening of the cis-Cyclobutene isomer. Explain the temperature effect, and identify the...
-
Thanasi has a business snowplowing. He uses a 2018 Ford Rapture with a snowplow outfitted to it. While plowing a driveway, he negligently ran it into the side of the neighbour's house. The damage to...
-
Mary was so excited about the holiday shopping season that she could hardly wait for the Black Friday sales. She got up early and spent all day at her favorite store, Mega Deals, shopping. Mary was...
-
This can be done as a group or individually. If done in a group, all group members must submit a copy of this assignment to receive a grade. If you were to open a business, what type of business...

Study smarter with the SolutionInn App


