Consider the following idealized PES spectrum for carbon: Explain the location and relative intensities of the various
Question:
Consider the following idealized PES spectrum for carbon:
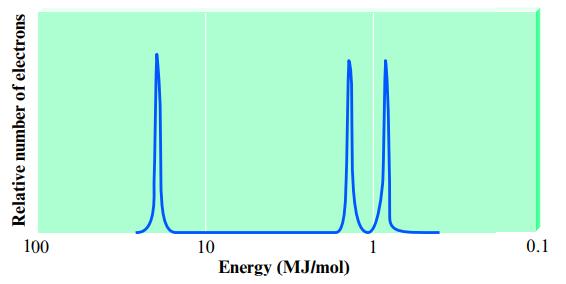
Explain the location and relative intensities of the various peaks.
Transcribed Image Text:
Relative number of electrons 100 10 Energy (MJ/mol) 1 0.1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
Carbon has an atomic number of 6 The electronic setup of it is Orbitals come in three main variet...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Consider the following idealized PES spectrum for potassium: Explain the location and relative intensities of the various peaks. Relative number of electrons 1000 100 10 Energy (MJ/mol) 0.1
-
Consider the following idealized PES spectrum for an element: What is the identity of the element? Explain the relative positions of the various peaks. Relative number of electrons 1000 100 t 1 10...
-
Consider the following idealized PES spectrum for an element: What is the identity of the element? Relative number of electrons 1000 100 10 Energy (MJ/mol) 0.1
-
Hatch plc has two classes of share capital outstanding: 8%, 20 par preference and 5 par ordinary. At December 31, 2021, the following accounts were included in equity. Share CapitalPreference,...
-
For a new product, sales volume in the first year is estimated to be 80,000 units and is projected to grow at a rate of 4% per year. The selling price is $ 12 and will increase by $ 0.50 each year....
-
On presenting your manager with the differential analysis of two possible uses for a piece of land that cost the company $1.5 million your manager believes you have made a major error as you omitted...
-
The stream function for an incompressible flow field is given by the equation \[ \psi=3 x^{2} y-y^{3} \] where the stream function has the units of \(\mathrm{m}^{2} / \mathrm{s}\) with \(x\) and...
-
Leila Durkin, an architect, opened an office on May 1, 2012. During the month, she completed the following transactions connected with her professional practice: a. Transferred cash from a personal...
-
A bond with a coupon rate of 12 percent sells at a yield to maturity of 14 percent. If the bond matures in 12 years, what is the Macaulay duration of the bond? What is the modified duration?
-
1. Where would you fit the preparation of a website for Taylor Stitch in their marketing plan? Why? 2. What metrics would you recommend to the owners of Taylor Stitch to help them determine if they...
-
Give a possible set of values of the four quantum numbers for all the electrons in a boron atom and a nitrogen atom if each is in the ground state.
-
How many 4d electrons would be predicted in the ground state for the following elements? a. Zirconium b. Cadmium c. Iridium d. Iron
-
How do we define current liabilities? Long-term liabilities?
-
You are buying a ( $ 2 0 , 0 0 0 ) boat with a down - payment of ( $ 4 , 0 0 0 ) and you finance the remaining amount with a 3 year loan at ( 7 . 8 % ) compounded weekly. ( A ) What are your weekly...
-
The mass of table sugar is 11.98 ounces (oz). Express its weight in gram-force if g = 31.66 ft/s. a. 335.14 gr W b. 336.14 gr c. 333.14 gf d. 334.14 gf
-
Suppose the annual yield to maturity on a 1 year zero coupon bond is 2 %, a 2 year zero coupon bond is 3 %, and a 3 year zero coupon bond is 4 %. (a) Consider a bond of face value 10,000 that pays a...
-
Combine and simplify. 20y 36 22y + 36
-
35. In 2005, Lee began working as a nurse anesthoist with a taxable income of $119,000. The Medicare tax was 1.45% and the Social Security tax rate was 6.2% to the maximum income of $90,000 that...
-
A miner working 260 m below sea level opened a carbonated soft drink during a lunch break. To his surprise, the soft drink tasted rather "flat." Shortly afterward, the miner took an elevator to the...
-
A new car sold for $31,000. If the vehicle loses 15% of its value each year, how much will it be worth after 10 years?
-
How would the energy versus dihedral angle plot for 2-methylpropane (isobutene) differs from that for propane?
-
Draw an energy versus dihedral angle plot for the conformations of 2, 3-dirnethylbutane about the C-2C-3 bond.
-
Discuss the geometry and the types of strain present in these compounds: (a) Cyclopropane (b) Cyclobutene (c) Cyclopentane (d) Cyclohexane (e) Cyclodecane
-
How do the resource descriptions (typing) used in the Incident Command System lead to a more effective response?
-
A company incurs $4172000 of overhead each year across three departments: Ordering and Receiving, Mixing, and Testing. The company prepares 2000 purchase orders, works 50000 mixing hours, and...
-
Hemming Company reported the following current-year purchases and sales for its only product. Date January 1 January 10 Activities Beginning inventory March 14 March 15 July 30 Sales Purchase Sales...

Study smarter with the SolutionInn App


