Consider the following mixture of SO 2 (g) and O 2 (g). If SO 2 (g) and
Question:
Consider the following mixture of SO2(g) and O2(g).
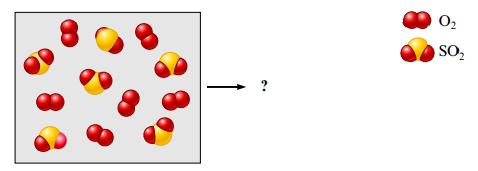
If SO2(g) and O2(g) react to form SO3(g), draw a representation of the product mixture assuming the reaction goes to completion. What is the limiting reactant in the reaction? If 96.0 g of SO2 react with 32.0 g O2, what mass of product will form?
Transcribed Image Text:
? 0₂ SO₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a The limiting reactan...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
What is a limiting reactant in a reaction mixture? Explain how it determines the amount of product.
-
What is a limiting reactant problem? Explain the method you are going to use to solve limiting reactant problems.
-
Describe how you could separate the following mixture of metal ions: Cd2+, Pb2+, and Sr2+.
-
The number x of bicycle helmets people are willing to buy per week from a retail chain at a price of $p is given by x = 1,000 - 60p + 25 20 ¤ p ¤ 100 (see the figure). (A) Find dx/dp....
-
Because expenses incurred both in a business and for the production of investment income are deductible, why is it important to determine in which category a particular activity falls?
-
Key financial figures for Best Buys fiscal year ended February 26, 2005, follow. Required 1. What is the total amount of assets invested in Best Buy? 2. What is Best Buys return on assets? Its assets...
-
In 2009 the United States was in a deep recession. What would be the expected effect of a recession on accounts receivable turnover ratios?
-
Gerald Pat Arrington was diagnosed with a brain tumor. At the time of the diagnosis, he was married to Brenda Arrington, but they were separated pending their divorce. Brenda and Pat had no children,...
-
A record company bought the rights to an artist's music catalogue and they expect to receive royalty payments of $30,000 per year forever (a perpetuity). What is this cash flow worth? Assume interest...
-
OCAS PROJECT. Effect of Damping. Consider a vibrating system of your choice modeled by y" + cy' + ky = d(t). %3D (a) Using graphs of the solution, describe the effect of continuously decreasing the...
-
You know that chemical A reacts with chemical B. You react 10.0 g A with 10.0 g B. What information do you need to determine the amount of product that will be produced? Explain.
-
Consider an iron bar on a balance as shown. As the iron bar rusts, which of the following is true? Explain your answer. a. The balance will read less than 75.0 g. b. The balance will read 75.0 g. c....
-
Which of the research methods discussed in this section would be best suited to study the impact of diet and exercise on the prevalence of a disease such as diabetes? Why?
-
Two machines were purchased in the financial year to 30 June 2022. Machine 1 is depreciated using the diminishing balance method at 35% p.a. It was purchased and installed on 31 January 2022 for $44...
-
Compute the taxable income for 2019 under each of the following circumstances: a. Jim is married and files a joint return. Jim and his wife have two dependent children. They have adjusted gross...
-
Quantitative Risk Assessment. Assume the following information: Calculate the overall risk factor for this project. Would you assess this level of risk as low, moderate, or high? Why? Probability of...
-
Go to http://tele-immersion.citris-uc.org/ and explore the nature of the project working to develop tele-immersion technology. Click the link marked Projects and observe the different fields and uses...
-
Access the Web site https://teamblue.unicomsi.com/products/focal-point/?cm_mc_uid=8153203956171487 3469682&cm_mc_sid_50200000=1487346968. What is IBMs philosophy regarding project portfolio...
-
Bramble University sells 3,500 season basketball tickets at $80 each for its 10-game home schedule. Give the entry to record (a) The sale of the season tickets and (b) The revenue recognized after...
-
The column shown in the figure is fixed at the base and free at the upper end. A compressive load P acts at the top of the column with an eccentricity e from the axis of the column. Beginning with...
-
Prove that the perfect gas temperature scale and the thermodynamic temperature scale based on the Second Law of thermodynamics differ from each other by at most a constant numerical factor.
-
Evaluate (ClS/ClV)]' for (a) A van der Waals gas, (b) A Dieterici gas (Table 1.7). For an isothermal expansion, for which kind of gas (and a perfect gas) will /).5be greatest? Explain your conclusion.
-
Two of the four Maxwell relations were derived in the text, but two were not. Complete their derivation by showing that (S/V)T = (p/T)V (T/P)s = (V/S)p
-
9 1. Compute the indefinite integral. +1 (3+ 4+4) dr.
-
Sarah Jackson Antiques issued its 11%, 10-year bonds payable at a price of $338,220 (face value is $400,000). The company uses the straight-line amortization method for the bond discount or premium....
-
Suppose you start with a full tank of gas (14 gallons) in your truck. After driving 6 hours, you now have 6 gallons left. If x is the number of hours you have been driving, then y is the number of...

Study smarter with the SolutionInn App


