Consider the following two acids: In two separate experiments the pH was measured during the titration of
Question:
Consider the following two acids:
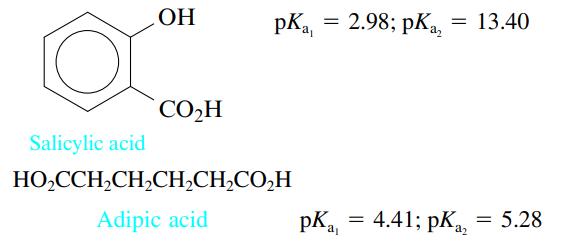
In two separate experiments the pH was measured during the titration of 5.00 mmol of each acid with 0.200 M NaOH. Each experiment showed only one stoichiometric point when the data were plotted. In one experiment the stoichiometric point was at 25.00 mL added NaOH, and in the other experiment the stoichiometric point was at 50.00 mL NaOH. Explain these results.
Transcribed Image Text:
OH CO₂H = pKa, = 2.98; pka₂ : 13.40 Salicylic acid HO₂CCH₂CH₂CH₂CH₂CO₂H Adipic acid pK₁ = 4.41; pKa a₂ 5.28
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (13 reviews)
Solution The results explanation entails an understanding of the concept of equivalence point ...View the full answer

Answered By

James Warinda
Hi! I’m James Otieno and I'm an experienced professional online tutor with countless hours of success in tutoring many subjects in different disciplines. Specifically, I have handled general management and general business as a tutor in Chegg, Help in Homework and Trans tutor accounts.
I believe that my experience has made me the perfect tutor for students of all ages, so I'm confident I can help you too with finding the solution to your problems. In addition, my approach is compatible with most educational methods and philosophies which means it will be easy for you to find a way in which we can work on things together. In addition, my long experience in the educational field has allowed me to develop a unique approach that is both productive and enjoyable.
I have tutored in course hero for quite some time and was among the top tutors awarded having high helpful rates and reviews. In addition, I have also been lucky enough to be nominated a finalist for the 2nd annual course hero award and the best tutor of the month in may 2022.
I will make sure that any student of yours will have an amazing time at learning with me, because I really care about helping people achieve their goals so if you don't have any worries or concerns whatsoever you should place your trust on me and let me help you get every single thing that you're looking for and more.
In my experience, I have observed that students tend to reach their potential in academics very easily when they are tutored by someone who is extremely dedicated to their academic career not just as a businessman but as a human being in general.
I have successfully tutored many students from different grades and from all sorts of backgrounds, so I'm confident I can help anyone find the solution to their problems and achieve
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Consider the following two independent situations: 1. A manufacturer reported an inventory turnover ratio of 8.6 during 2013. During 2014, management introduced a new inventory control system that...
-
Consider the following two samples: Sample 1: 10, 9, 8, 7, 8, 6, 10, 6 Sample 2: 10, 6, 10, 6, 8, 10, 8, 6 (a) Calculate the sample range for both samples. Would you conclude that both samples...
-
Consider the following two mutually exclusive projects: Whichever project you choose, if any, you require a 15 percent return on your investment. (a) If you apply the payback criterion, which...
-
3. (15 pts.) The random variables X and Y have joint density function fxx(x, y) x-?y?, x > 1, y > 1. Compute the cdf and pdf of the random variable U = XY.
-
Art Wyatt Pool Company wishes to finance a $15 million expansion program and is trying to decide between debt and external equity. Management believes that the market does not appreciate the...
-
Consider a U-tube whose arms are open to the atmosphere. Now equal volumes of water and light oil (r = 49.3 lbm/ft3) are poured from different arms. A person blows from the oil side of the U-tube...
-
You roll a die. Find the probability of each event. (a) Rolling a 6 or a number greater than 4 (b) Rolling a 2 or a prime number (c) Rolling a number less than 5 or an odd number
-
I know that its the thing to do, insisted Pamela Kincaid, vice president of finance for Colgate Manufacturing. If we are going to be competitive, we need to build this completely automated plant. Im...
-
Calculate the mean, mode, median, standard deviation, and range for the Graduation Rate, Salary After Completing Program, and Annual Cost. our response to each of these should include only the mean,...
-
Consider the project data model shown in Figure 1-15. a. Create a textual description of the diagrammatic representation shown in the figure. Ensure that the description captures the...
-
The common ion effect for weak acids is to significantly decrease the dissociation of the acid in water. Explain the common ion effect.
-
Sketch the titration curves for a diprotic acid titrated by a strong base and a triprotic acid titrated by a strong base. List the major species present at various points in each curve. In each...
-
For the year ended December 31, Global Exports had net sales of $7,750,000, costs and other expenses (including income tax) of $6,200,000, and an extraordinary gain (net of income tax) of $420,000....
-
Question 1 The following table gives the income and expenses for a UNLV student. Calculate the monthly cash flow. Income College Savings (per year) = $26,400 O $9,650 O -$90 O $804.17 O-$1080...
-
Which qualitative characteristic or assumption is best represented by the following statement? Businesses will continue their operations into the future. 1) separate entity 2) unit-of-measure 3)...
-
This bid document is used for complex or complicated scope where the buyer is looking for the vendor to provide a solution. A. Request for quote B. Request for information C. Request for proposal D....
-
Item4 Time Remaining 17 minutes 22 seconds 00:17:22 eBook Item 4 Time Remaining 17 minutes 22 seconds 00:17:22 Those firms that make relatively conservative estimates for their accrued and deferred...
-
When Juan Hernandez was first given the position of head buyer for the Connery Manufacturing Company, he visualized the job as merely an expansion of his old position as a commodity buyer. He had no...
-
Discuss the main reasons for the losses of the U.S. savings and loans industry in the 1980s.
-
In the operation of an automated production line with storage buffers, what does it mean if a buffer is nearly always empty or nearly always full?
-
Assume you are carrying out the dehydration of 1-methylcyclohexanol to yield 1-methykyclohexene. How could you use infrared spectroscopy to determine when the reaction is complete?
-
Assume that you are carrying out the base-induced de-hydro bromination of 3-bromo-3-methylpentane (Section 11.7) to yield an alkene. How could you use IR spectroscopy to tell which of two possible...
-
Which is stronger, the C = O bond in an ester (1735 cm1) or the C = O bond in a saturated ketone (1715 cm1)? Explain.
-
"Managing Away Bad Habits Team Assignment Organizational Behavior IILeadership Assigned is ashort case from the exercise Managing Away Bad Habits. The task is to develop a turnaround strategy for...
-
"Managing Away Bad Habits Team Assignment Organizational Behavior IILeadership Assigned is ashort case from the exercise Managing Away Bad Habits. The task is to develop a turnaround strategy for...
-
11 The APRN unit director is working at an agency that has a high incidence of medication errors, specifically with heparin. One recent error had a patient receive twice the prescribed dose. Which...

Study smarter with the SolutionInn App


