Consider two separate gas containers at the following conditions: How is the pressure in container B related
Question:
Consider two separate gas containers at the following conditions:
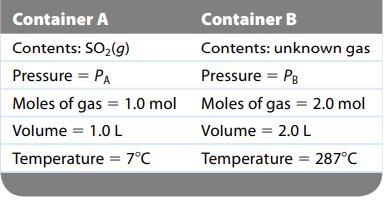
How is the pressure in container B related to the pressure in container A?
Transcribed Image Text:
Container A Contents: SO₂(g) Pressure = PA Container B Contents: unknown gas Pressure = PB of gas Moles of gas Volume = 2.0 L Temperature = 287°C Moles of gas = 1.0 mol Moles Volume = 1.0 L Temperature = 7°C = 2.0 mol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 30% (10 reviews)
They are related by a ratio The pressure in container B is twice the pressure as in container A So ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Use the definition of the definite integral to justify the property where f is continuous and c is a real number. x = c [" f(x) dx, S"cf(x) dx a
-
A gas in a container had a measured pressure of 57 kPa. Calculate the pressure in units of atm and mmHg.
-
Consider the following gas container equipped with a movable piston. a. By what factor (increase by 1, decrease by 1.5, etc.) would you change the pressure if you wanted the volume to change from...
-
Research about the competitive and comparative advantage of the Argentina in terms of physical and human resources and how it is making use of these resources for international trade purpose. 3....
-
Matilda and John Perry owned property that was damaged by fire. The Perrys were insured against property damage caused by fire under an insurance policy issued by AFRD Insurance Company of Canada....
-
Find the derivative of the function. Simplify where possible. y = arctan1 - x/1 + x
-
Determine the largest length of a structural A-36 steel rod if it is fixed supported and subjected to an axial load of \(100 \mathrm{kN}\). The rod has a diameter of \(50 \mathrm{~mm}\). Use the AISC...
-
This Mini Case incorporates the calculation of a weighted average cost of capital (WACC) to determine whether the Alpha One Software Corporation added economic value in 2010. The Appendix to Chapter...
-
1. A 115 volt fluorescent lamp is rated at 0.43 amps (430 milliamperes). Find the watts that will be consumed by the lamp and its ballast. (115 volts is specified instead of 120 volts to allow for...
-
There is considerable strain in forming a flat ring so it is only adopted when there is some sort of stabilization that outweighs this. This extra stability is aromaticity. From your research on the...
-
Concentrated hydrogen peroxide solutions are explosively decomposed by traces of transition metal ions (such as Mn or Fe): What volume of pure O 2 (g), collected at 27 C and 746 torr, would be...
-
A 5.0-L flask contains 0.60 g O 2 at a temperature of 22 C. What is the pressure (in atm) inside the flask?
-
An aerial photographic system is used for stereophotography. The film negative is a square of sides, the focal length is f, the baseline is band the photographs (which are acquired with the camera...
-
Agree with this post: The Games approach offers additional advantages, such as fostering a sense of teamwork and decision - making skills as players navigate dynamic game scenarios. Furthermore, it...
-
The 1 9 7 0 s allow listeners more personal chores and what they listen to at home and in their cars later people gain even more freedom when they could listen in their portable headphones when they...
-
Look back at the results that you got for Part A and Part B of the Individual Assignment on Paid Search Bid Optimization. You should notice that across the four keywords, there is a relationship...
-
What is troublesome to you when communicating or interacting with other people? What do you have the most difficulty with in group situations? Are you better talker or listener? ( Circle ) Explain:...
-
Here is a diagram of a acute triangle. What is the lenght of side b (express your answer to the nearest tenth) B 16 $20 20 b My answer is (1 decimal place) is: 1 Question 2 (1 point) For this acute...
-
Let L be the linear operator on R2 defined by L(x) = (x1) cos - x2 sin , x1 sin + x2 cos )T Express x1, x2, and L(x) in terms of polar coordinates. Describe geometrically the effect of the linear...
-
Stephen Schor, an accountant in New York City, advised his client, Andre Romanelli, Inc., to open an account at J. P. Morgan Chase Bank, N.A., to obtain a favorable interest rate on a line of credit....
-
Determine the electron configuration for each of the following atoms: a. Carbon b. Oxygen c. Boron d. Fluorine e. Sodium f. Aluminum
-
In each case, identify the more stable anion. Explain why it is more stable. (a) (b) (c) vs. N. vs. -zo
-
Atropine, extracted from the plant Atropa belladonna, has been used in the treatment of bradycardia (low heart rate) and cardiac arrest. Draw the enantiomer of atropine: CH 0= -
-
Suresh Company reports the following segment (department) income results for the year. Sales: Expenses Avoidable Unavoidable Total expenses Income (loss) Department M $ 66,000 Department N $ 38,000...
-
EZ Tees Corporation produces T-shirts. The company uses a standard costing system and has set the following standards for direct materials and direct labor (for one shirt): Fabric (1.5 yds. @ $2.80)...
-
Question : List the competitive advantages of Nordstrom: the things that make it different from competitors in positive ways. Nordstrom: A Shining Example of Service Excellence 'Use your best...

Study smarter with the SolutionInn App


