Determine the phases that are present and the compositions for each phase in Cu-85 wt% Ag at
Question:
Determine the phases that are present and the compositions for each phase in Cu-85 wt% Ag at 800°C.
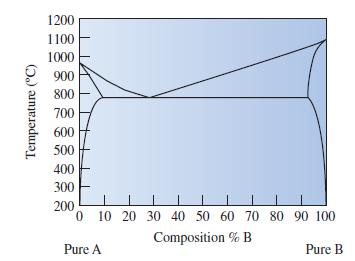
Transcribed Image Text:
Temperature (°C) 1200 1100 1000 900 800 700 600 500 400 300 200 0 10 20 30 40 50 60 70 80 90 100 Composition % B Pure A Pure B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
ANSWER At 800 C the phases present are liquid an...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Determine the phases that are present and the compositions for each phase in Cu-55 wt% Ag at 600C. Data Form Problem 11-23 Consider a Pb-70% Sn alloy. Determine (a) If the alloy is hypoeutectic or...
-
Cite the phases that are present and the phase compositions for the following alloys: (a) 90 wt% Zn-10 wt% Cu at 400(C (750(F) (b) 75 wt% Sn-25 wt% Pb at 175(C (345(F) (c) 55 wt% Ag-45 wt% Cu at...
-
Cite the phases that are present and the phase compositions for the following alloys: (a) 90 wt% Zn-10 wt% Cu at 400C (750F) (b) 75 wt% Sn-25 wt% Pb at 175C (345F) (c) 55 wt% Ag-45 wt% Cu at 900C...
-
Consider a static (one-period), closed economy with one representative consumer, one rep- resentative firm, and a government. The level of capital K and government expenditures G in the economy are...
-
For the given of Problem 11, indicate: (a) How much would an interest arbitrageur earn if the foreign currency were at a forward premium of 1 percent per year? (b) What would happen if the foreign...
-
For what values of p does the series converge? For what values of p does it diverge? kP k=1
-
What is an Organizational System?
-
A tank in a room at 19C is initially open to the atmosphere on a day when the barometric pressure is 102 kPa. A block of dry ice (solid CO2) with a mass of 15.7 kg is dropped into the tank, which is...
-
The contribution margin ratio of Kuck Corporation's only product is 62%. The company's monthly fixed expense is $455,000 and the company's monthly target profit is $41,000. Required: Determine the...
-
Hot Spot is a firm based in the United States whose core business is the maintenance of home heaters. The company usually forecasts service requests on the basis of the number of installed heaters....
-
A Pb-Sn alloy contains 45% and 55% at 100C. Determine the composition of the alloy. Is the alloy hypoeutectic or hypereutectic?
-
A Pb-Sn alloy contains 23% primary a and 77% eutectic microconstituent immediately after the eutectic reaction has been completed. Determine the composition of the alloy.
-
A constant-pressure piston/cylinder contains 0.5 kg air at 300 K, 400 kPa. Assume the piston/ cylinder has a total mass of 1 kg steel and is at the same temperature as the air at any time. The system...
-
Interest versus dividend expense Michaels Corporation expects earnings before interest and taxes to be $40,000 for the current period. Assuming a flat ordinary tax rate of 21%, compute the firm's...
-
Garth is married to Hana, a native and citizen of Norway. Garth is a U.S. citizen. They live in New York City. He owns a life insurance policy on his life. The latest statement from the life...
-
The Manning Company has financial statements as shown next, which are representative of the company's historical average. The firm is expecting a 30 percent increase in sales next year, and...
-
An investment will pay $50 at the end of each of the next 3 years, $200 at the end of Year 4, $400 at the end of Year 5, and $550 at the end of Year 6. If other investments of equal risk earn 12%...
-
Consider two mutually exclusive projects A and B: Project Co Cash Flows (dollars) G C NPV at 10% A -33,000 23,400 B -53,000 36,000 23,400 36,000 +$ 7,612 +9,479 a. Calculate IRRS for A and B. Note:...
-
Assuming that the force constant is approximately the same for C--C, C--N, and C--O bonds, predict the relative positions of their stretching vibrations.
-
Interest Compounded Annually. When P dollars is invested at interest rate i, compounded annually, for t years, the investment grows to A dollars, where A = P(1 + i) t . Trevor's parents deposit $7800...
-
(a) Using only K sp from Table 6-3, calculate how many moles of Ca(OH) 2 will dissolve in 1.00 L of water. (b) How will the solubility calculated in part (a) be affected by the K 1 reaction in Table...
-
From the following equilibrium constants, calculate the equilibrium constant for the reaction HO,CCO,H 2H* + C,0.
-
Assuming complete dissociation of the salts, calculate the ionic strength of (a) 0.2 mM KNO 3 ; (b) 0.2 mM Cs 2 CrO 4 ; (c) 0.2 mM MgCl 2 plus 0.3 mM AlCl 3 .
-
Pietro Frozen Foods, Inc., produces frozen pizzas. For next year, Pietro predicts that 50,000 units will be produced, with the following total costs: Direct materials Direct labor Variable overhead...
-
You will complete two case studies over the course of the semester. These are to be completed individually. Each case study is found in the textbook and there are questions there to help guide your...
-
Thanasi has a business snowplowing. He uses a 2018 Ford Rapture with a snowplow outfitted to it. While plowing a driveway, he negligently ran it into the side of the neighbour's house. The damage to...

Study smarter with the SolutionInn App


