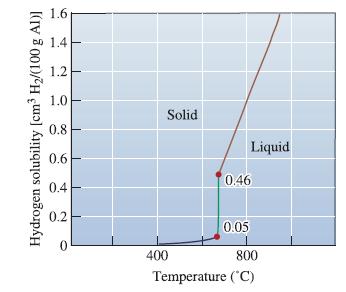
From Figure 9-14, find the solubility of hydrogen in liquid aluminum just before solidification begins when the
Question:
From Figure 9-14, find the solubility of hydrogen in liquid aluminum just before solidification begins when the partial pressure of hydrogen is 1 atm. Determine the solubility of hydrogen (in cm3/100 g Al) at the same temperature if the partial pressure were reduced to 0.01 atm.
Transcribed Image Text:
Hydrogen solubility [cm³ H₂/(100 g Al)] 1.6 1.4 1.2 1.0 0.8 0.6 0.4 0.2 Solid 400 Liquid 0.46 0.05 800 Temperature (°C)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
The solubility of hydrogen in cm3100 g Al 3365 00677 x 1 atm 00117 ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
The solubility of hydrogen in liquid aluminum at 715C is found to be 1 cm 3 / (100 g Al). If all of this hydrogen precipitated as gas bubbles during solidification and remained trapped in the...
-
Repeat Prob. 965E if the compression ratio were reduced to 12. Data From Q#65: An air-standard dual cycle has a compression ratio of 20 and a cutoff ratio of 1.3. The pressure ratio during the...
-
The solubility of hydrogen gas in steel in terms of its mass fraction is given as WH, = 2.09 X 104 exp(-3950/T)P p05 where PH2 is the partial pressure of hydrogen in bars and T is the temperature in...
-
Suppose that a firm is producing in the short run with output given by: Q = 200.5L 2.5L 2 , The firm hires labor at a wage of $25 per hour and sells the good in a competitive market at P = $50 per...
-
Find the degree of intra-industry trade if exports and imports are, respectively (a) 1,000 and 1,000 (b) 1,000 and 750 (c) 1,000 and 500 (d) 1,000 and 25 (e) 1,000 and 0
-
Suppose a company you are considering as an investment made sales of \(\$ 54.8\) billion in the year ended December 31, 2020. Collections from customers totalled \(\$ 55\) billion. The company began...
-
What is the purpose of a declaration?
-
Equivalence of various forms of Fick's law for binary mixtures (a) Starting with Eq. (A) of Table 17.8-2, derive Eqs. (B), (D), and (F). (b) Starting with Eq. (A) of Table 17.8-2, derive the folowing...
-
Have an SQL Query that will pullout the Error Rate Summary per TL (Team Lead) THe Google sheet link is attached here....
-
Outline practical syntheses of each of the following compounds from alcohols containing no more than four carbon atoms and any necessary organic or inorganic reagents. In many cases the desired...
-
Explain the green sand molding process.
-
Why is it that castings made from pressure die casting are likely to be stronger than those made using the sand casting process?
-
When oil (kinematic viscosity \(1 \times 10^{-4} \mathrm{~m}^{2} / \mathrm{s}\), specific gravity 0.92) flows at a mean velocity of \(1.5 \mathrm{~m} / \mathrm{s}\) through a 50 -mm-diameter...
-
What are the basic forms of systems model? Give an example of each and their role in a project environment.
-
Why is the subject of organizational structure so important?
-
Why is making progress visible so important?
-
Briefly list the other ways in which an organization may be structured.
-
What is monitoring and control in the context of project management?
-
Refer to E4-3 and E4-5. E4-3 Diane Company completed its first year of operations on December 31. All of the year's entries have been recorded except for the following: a. At year-end, employees...
-
What will be the final value of DI after executing the following piece of code? Execute the instructions dependently one after another. CLD MOU CX,OFOH MOU AX.02874H MOU DI,01000H MOU ES, DI SUB...
-
Give three examples of engineered products that are designed to work well over a million times.
-
Select a type of product that would have versions in all four quadrants in the style-versus-technology design chart (see Focus On Innovation). Show the four versions of the product, and clearly...
-
Imagine you are tasked with designing a coffee maker that would be marketable to cafs around the globe. Conduct research on coffee makers to determine a set of global, social, environmental, and...
-
You are planning to retire in 30 years. You want to be able to spend $40,000 per year in retirement, adjusted for inflation (so you will spend the equivalent in each year of $40,000 in today's...
-
From a lawsuit, you have been awarded a 31-payment, constant growth annuity. The first payment is at Year O and is equal to $380, and each subsequent payment will be paid in 16 month intervals, with...
-
You will receive 13 payments of $535, where the first payment will be received today (Month 0) and all other payments will be received in 10-month intervals (Months 10, 20, 30 ... 120). Assume that...

Study smarter with the SolutionInn App


