Give the maximum number of electrons in an atom that can have these quantum numbers: a. n
Question:
Give the maximum number of electrons in an atom that can have these quantum numbers:
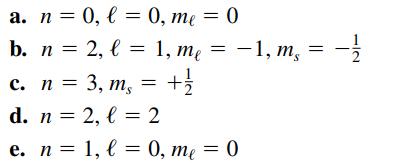
Transcribed Image Text:
a. n = 0, l= 0, me = b. n = 2, l = 1, me 3, ms = + 1/2 c. n = d. n = 2, l = 2 e. n = 1, l = 0, me = 0 0 -1, ms --/12
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
The maximum number of electrons having ...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
What is the maximum number of electrons in an atom that can have these quantum numbers? a. n = 4 b. n = 5, m = +1 c. n = 5, ms = + 1/2 d. n = 3, = 2 e. n = 2, = 1 f. n = 0, = 0, m = 0 g. n = 2, =...
-
What is the maximum number of electrons in an atom that can have the following quantum numbers? Specify the orbitals in which the electrons would be found. (a) n = 2, ms = + 1/2; (b) n = 4, m = 11;...
-
What is the maximum number of electrons in an atom that can have the following quantum numbers: (a) n = 2, m s = -1/2, (b) n = 5, l = 3, (c) n = 4, l = 3, m 1 = 3; (d) n = 4, l = 0, m 1 = 0?
-
The mid-day wind speed, in knots, at a coastal resort is normally distributed with mean 12.8 and standard deviation . a. Given that 15% of the recorded wind speeds are less than 10 knots, find the...
-
MasterTech is a new software company that develops and markets productivity software for municipal government applications. In developing their income statement, the following formulas are used:...
-
Suppose X ~ N(12, 6). What is the z-score of x = 2?
-
The adjusting entries for the following adjustments were omitted at year-end: a. Prepaid insurance expired, $2,400. b. Depreciation, $1,800. c. Employee salaries owed for Monday through Wednesday of...
-
Assume that a 1.00-kg ball is thrown solely by the action of the forearm, which rotates about the elbow joint under the action of the triceps muscle, Fig. 8-45. The ball is accelerated uniformly from...
-
The comparative financial statements prepared at December 31, year 2, for Goldfish Company showed the following summarized data: Statement of Earnings Sales revenue Cost of sales Gross profit...
-
What type of professional service engagement was the assignment completed by the team of auditors led by Katelyn Light? Defend your answer. Which professional standards are applicable to such...
-
In defining the sizes of orbitals, why must we use an arbitrary value, such as 90% of the probability of finding an electron in that region?
-
In the 1930s, electron microscopes were first used to provide structural details of human cells. If an electron microscope uses electrons having a wavelength of 0.1 nm, calculate the velocity of the...
-
Refer to problem P12-45. Assume that SCMI elects to report its financial results in accordance with Accounting Standards for Private Enterprises (ASPE). Problem P12-45 During 2021, Surinders Copper...
-
Water is flowing in a horizontal pipe bend at a velocity of $10 \mathrm{ft} / \mathrm{s}$. The radius of curvature of the inside of the bend is 4 in., and the pipe ID is 2 in. A mercury manometer is...
-
An orifice having a diameter of $1 \mathrm{in}$. is used to measure the flow rate of SAE 10 lube oil $(\mathrm{SG}=0.928)$ in a $2 \mathrm{in}$. sch 40 pipe at $70^{\circ} \mathrm{F}$. The pressure...
-
It is possible to achieve a weightless condition for a limited time in an airplane by flying in a circular arc above the earth (like a rainbow). If the plane flies at $650 \mathrm{mph}$, what should...
-
A 2 in. sch 40 pipe carries a $35^{\circ} \mathrm{API}$ distillate at $50^{\circ} \mathrm{F}(\mathrm{SG}=0.85)$. The flow rate is measured by an orifice meter, which has a diameter of $1.5...
-
A 4 in. sch 80 pipe carries water from a storage tank on top of a hill to a plant at the bottom of the hill. The pipe is inclined at an angle of $20^{\circ}$ to the horizontal. An orifice meter with...
-
Assume Spahr and Kennedy, the accounting firm, advises Arctic Seafood that its financial statement must be changed to conform to GAAP. At December 31, 2016, Arctic's accounts include the following:...
-
Find the velocity, acceleration, and speed of a particle with the given position function. r(t) = (t 2 , sin t - t cos t, cos t + t sin t), t > 0
-
Build a handheld model of bicycle [2.2.1] heptanes and discuss the types of strain that are present in this compound. udy tee op!
-
Explain whether the compound shown is the Z or the E diastereomer. Problems using online Three-Dimensional molecular models
-
Explain whether the conformation shown is the most stable conformation of each of these molecules. Problems using online Three-Dimensional molecular models
-
This is for my Organizational Behavior class. This is due by midnight tonight, July 30. Please help!! The organization I have chosen is a rural health care facility...a clinic. What are the basic...
-
What are the functions of organizational culture? Use your own experience in an organization (workplace, school, gym, etc.) to explain how organizational culture contributes either positively or...
-
In 2 0 2 4 , Bratten Fitness Company made the following cash purchases: The exclusive right to manufacture and sell the X - Core workout equipment from Symmetry Corporation for $ 2 1 8 , 0 0 0 ....

Study smarter with the SolutionInn App


