Given the following data: calculate G for the reaction 2CHl) + 15O,(g) C(s) + O(g) H(g)
Question:
Given the following data:
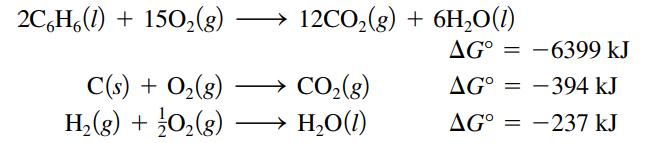 calculate ΔGο for the reaction
calculate ΔGο for the reaction
![]()
Transcribed Image Text:
2C¢Hl) + 15O,(g) C(s) + O(g) H»(g) + }O»(g) 12CO,(g) + 6H2O(l) → CO2(g) H2Ol) AG° = -6399 kJ = AG° = -394 kJ AG° = -237 kJ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
ANSWER To calculate the standard Gibbs free energy change G for the reaction 6Cs 3H2g C6H...View the full answer

Answered By

Churchil Mino
I have been a tutor for 2 years and have experience working with students of all ages and abilities. I am comfortable working with students one-on-one or in small groups, and am able to adapt my teaching style to meet the needs of each individual. I am patient and supportive, and my goal is to help my students succeed.
I have a strong background in math and science, and have tutored students in these subjects at all levels, from elementary school to college. I have also helped students prepare for standardized tests such as the SAT and ACT. In addition to academic tutoring, I have also worked as a swim coach and a camp counselor, and have experience working with children with special needs.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Given the following data from three in de pen dent samples, use the 0.025 level in determining whether the population means could be the same. (Use data file XR12092.) 3-48377 11212 701536 21121 2...
-
Given the following data on inputs and outputs at a work center, determine the cumulative deviation and the backlog for each time period. The beginning backlog is7. PERIOD Input Planned2 Actua2 200...
-
Given the following data from group 1 and group 2: a. Combine the two datasets in ranked order. Compute the sum of the ranks for group 1, R1. Compute the sum of the ranks for group 2, R2. b. Compute...
-
It was reported that 23% of U.S. adult cellphone owners called a friend for advice about a purchase while in a store. If a sample of 15 U.S adult cellphone owners is selected, what is the probability...
-
Suppose the discount rate is below the federal funds rate, and banks can borrow as much as they want from the Fed. How could a bank earn easy profits? Would the federal funds rate stay above the...
-
In some wind tunnels the test section is perforated to suck out fluid and provide a thin viscous boundary layer. The test section wall in Fig P3.33 contains 1200 holes of 5-mm diameter each per...
-
Find a case in California that discusses discovery abuse. What was the abuse? Were there sanctions given by the court against one party for the abuse?
-
On November 1, 2019, the account balances of Schilling Equipment Repair were as follows. During November, the following summary transactions were completed. Nov. 8 Paid $1,700 for salaries due...
-
use the course content ofHuman Behavior in Businessto answer questions ABC Corporation is a small enterprise with 100 employees and four managers. John and Jane are co-workers who got along well with...
-
Danny Imasuen is a 37-year-old student working in Quebec. His wages for the current weekly pay period are $580.00. The employer pays $22.00 for life insurance premiums and $190.00 for group medical...
-
For the sublimation of iodine at 25C the values of H o and G o are, respectively, 62 kJ and 19 kJ. Estimate the temperature at which iodine sublimes. Assume H o and S o do not depend on temperature....
-
Calculate H o , S o , and G o at 25C for each of the following reactions that occur in the atmosphere. a. CH4(g) + O2(g) CH3CHO(g) + O(g) b. 03(g) + NO(g) NO(g) + O(g) c. SO3(g) + HO(l) HSO4(aq)
-
USA Today reported (Anthony Breznican and Gary Strauss, Where Have All the Moviegoers Gone? June 23, 2005) that in the summer of 2005, ticket sales to movie theaters had fallen for 17 straight weeks,...
-
Fill in the Blank. The forced-vibration solution of Duffing's equation has _____________ for any given amplitude \(|A|\).
-
Fill in the Blank. The phenomenon of realizing two amplitudes for the same frequency is known as ____________ phenomenon.
-
If the rate of change of force with respect to displacement, \(d f / d x\), is an increasing function of \(x\), the spring is called a a. soft spring b. hard spring c. linear spring
-
A nonlinear stress-strain curve can lead to nonlinearity of the a. mass b. spring c. damper
-
Each term in the equation of motion of a linear system involves displacement, velocity, and acceleration of the a. first degree b. second degree c. zero degree
-
What effects does friction have on a forming-limit diagram? Explain.
-
To help you become familiar with the accounting standards, this case is designed to take you to the FASBs Web site and have you access various publications. Access the FASBs Web site at...
-
1, 3, 5-Hexatriene has max = 258 nm. In light of your answer to Problem 14.46, approximately where would you expect 2, 3-dimethyl-1, 3, 5-hexatriene to absorb?
-
?-Ocimene is a pleasant-smelling hydrocarbon found in the leaves of certain herbs. It has the molecular formula C10H16 and a UV absorption maximum at 232 nm. On hydrogenation with a palladium...
-
Myrcene, C10H16, is found in oil of bay leaves and is isomeric with ?-Ocimene (Problem 14.48). It has an ultraviolet absorption at 226 nm and can be catalytically hydrogenated to yield 2,...
-
"Paula is a social worker in a fostercare agency. She has been working with 9-year-old Rita, her foster mother, Flo, and biological mother, Stella, for three years. Rita was removed from her mother's...
-
You are 22 years old and currently have $100,000 in savings for retirement. You will start making annual contributions to your savings at the beginning of each year in the amount of $2,500. If you...
-
You have an investment opportunity that pays $500 every three months for 7 years and an additional $14,000 at the end of the seventh year. How much is the investment worth today if you can invest in...

Study smarter with the SolutionInn App


