Given the following data: Calculate the bond energy for the O 2 bond, that is, calculate H
Question:
Given the following data:
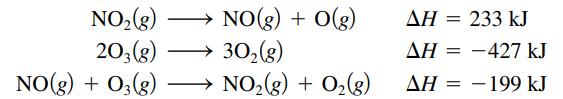
Calculate the bond energy for the O2 bond, that is, calculate ΔH for:
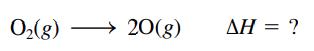
Transcribed Image Text:
NO2(g) 203(8) NO(g) + O3(g) - NO(g) + O(g) 30₂(g) NO,(g) + O2(g) ΔΗ = 233 kJ ΔΗ = −427 kJ = ΔΗ = – 199 kJ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
The reaction youve provided is O2g 2Og To calculate th...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Given the network in figure sketch the magnitude characteristic of the transfer function, identify the type offilter. "J) 1000 .F 100 ,() ) 10 H
-
Given the following data on inputs and outputs at a work center, determine the cumulative deviation and the backlog for each time period. The beginning backlog is7. PERIOD Input Planned2 Actua2 200...
-
Given the following data from three in de pen dent samples, use the 0.025 level in determining whether the population means could be the same. (Use data file XR12092.) 3-48377 11212 701536 21121 2...
-
For show by the method described in this chapter that the following pairs of FAs are equivalent: FA +1 (1 (1 a. b FA a b
-
Construct a scatter plot matrix for Median Income, Median Home Value, Median Household Wealth, and Average Bank Balance in the Excel file Banking Data. What conclusions can you reach?
-
The cost of unleaded gasoline in the Bay Area once followed an unknown distribution with a mean of $4.59 and a standard deviation of $0.10. Sixteen gas stations from the Bay Area are randomly chosen....
-
Use the Hillcrest Corporation data in Short Exercise 13-6 to compute Hilkest's a. Total liabilities b. Total assets Exercise 13-6 Hillcrest Corporation reported the following accounts: Prepare the...
-
One of New Centurys most important accounts was its loan repurchase loss reserve. Each accounting period, New Century was required to estimate the ending balance of that account. What general...
-
Consider a 3-month put option. Suppose that the underlying stock price is $25, the strike $26, the interest rate is 5% p.a., stock volatility is 6% per month. Use the same data to answer questions a)...
-
This alphabetized adjusted trial balance is for GalaVu Entertainment as of its December 31, 2023, year-end: Required Use the information in the trial balance to prepare: a. The income statement for...
-
Given the following data calculate H for the reaction Fe2O3(s) + 3CO(g) 3Fe,O,(s) + CO(g) Fe3O4(s) + CO(g) 2Fe(s) + 3CO2(g) 2Fe,O4(s) + CO2(g) 3FeO(s) + CO2(g) = = -23 kJ -39 kJ 18 kJ
-
In a coffee-cup calorimeter, 1.60 g NH 4 NO 3 is mixed with 75.0 g water at an initial temperature of 25.00 C. After dissolution of the salt, the final temperature of the calorimeter contents is...
-
On January l of this year, Cunningham Corporation issued bonds with a face value of $200,000 and a coupon rate of 6 percent. The bonds mature in 10 years and pay interest annually every December 31....
-
A limited company wants to change its method of providing for depreciation from reducing balance method to fixed instalment method. Can the company do so? If so, explain as to how? In this context,...
-
Is it absolutely necessary that depreciation should be provided for before profits are distributed as dividend? What are the provisions of the Companies Act, 2013, regarding the quantum of...
-
What are the major differences between ordinal and interval scales? In your response include an example of each type of scale.
-
Is depreciation on wasting assets a necessary charge for arriving at the true and fair picture of the financial statements of a company?
-
What are the critical issues involved in pretesting a questionnaire? How does a pretest differ from a pilot test?
-
The cash data of Duffy Automotive for July 2016 follow: Duffy Automotive received the following bank statement on July 31, 2016: Additional data for the bank reconciliation include the following: a....
-
A city maintains a solid waste landfill that was 12 percent filled at the end of Year 1 and 26 percent filled at the end of Year 2. During those periods, the government estimated that total closure...
-
Show the products of thesereactions: CH3 Br CH,CH,OH CH,OH a) Ph- -Br b) Ph CH; CH, Br . CH;OH Br d) ELOH Ph
-
Show the products of thesereactions: Br Br O NaOH NAOH b) PHCHCHCCH3 a)
-
Show the substitution and/or elimination products for these reactions. Explain which mechanism are occurring and which product you expect to be the majorone. OTs a) CH,CH,CHCH, + CH,CH,CO CH,CH,CO,H...
-
I am working in aMobile company that is seeking to introduce a novel mobile application that implements a monthly subscription fee for its users. After conducting a comprehensive analysis, the...
-
Tatiana has recently entered a profile into the Express Entry (EE) system as a candidate under the Canadian Experience Class (CEC). She has also met the requirements to qualify under the FSW. Tatiana...
-
Columbia Corp. is a young, rapidly growing company that manufactures computer accessories and specialized components for networked computer workstations. It has some unique products and a strong...

Study smarter with the SolutionInn App


