Iron oxide ores, commonly a mixture of FeO and Fe 2 O 3 , are given the
Question:
Iron oxide ores, commonly a mixture of FeO and Fe2O3, are given the general formula Fe3O4. They yield elemental iron when heated to a very high temperature with either carbon monoxide or elemental hydrogen. Balance the following equations for these processes:
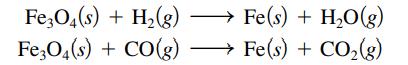
Transcribed Image Text:
Fe3O4(s) + H₂(g) Fe3O4(s) + CO(g) Fe(s) + H₂O(g) Fe(s) + CO₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
Fe3O4s 4H2g Fes 4H2Og Fe3O4s 3COg Fes 3CO2g The above equations are balanced ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
As a body is heated to a very high temperature and becomes self luminous, the apparent color of the emitted radiation shifts from red to yellow and finally to blue as the temperature increases. Why...
-
Moist air is heated to a very high temperature. If the equilibrium composition consists of H2O, O2, N2, OH, H2, and NO, the number of equilibrium constant relations needed to determine the...
-
A mixture of Fe2O3 and FeO was found to contain 72.00% Fe by mass. What is the mass of Fe2O3 in 0.500 g of this mixture?
-
Give a recursive algorithm for finding the reversal of a bit string. (See the definition of the reversal of a bit string in the preamble of Exercise 34 in Section 5.3.)
-
Tim retired during the current year at age 58. He purchased an annuity from American National Life Company for $40,000. The annuity pays Tim $500 per month for life. a. Compute Tims annual exclusion....
-
In the Equity Funding case all of the following were factors in the case except a. The integrity of management. b. Massive collusion by management. c. Liability for ordinary negligence. d. A...
-
Identify each of the following as land (L) or land improvements (LI): 1. Survey fees 2. Fencing 3. Lighting 4. Clearing land 5. Parking lot
-
On January 1, 2014, Yellow Car Company issued 15-year, $50,000,000 face value, 4% bonds, at par. Each $1,000 bond is convertible into 20 shares of Yellow Car common stock . None of the bonds were...
-
Wendy Cheung was at it again! It seemed like she was causing troubles wherever she went. At her previous job, Wendy had successfully persuaded the top management to implement a system that recognizes...
-
Q1. Use the LIFO cost-flow assumption to answer the following questions. YEAR 1: Purchase #1 1,000 units @ $1 = $1,000 Purchase #2 1,000 units @ $1 = $1,000 a. How much is goods available for sale?...
-
Balance each of the following chemical equations. a. KO (s) + HO(1)KOH(aq) + O(g) + HO(aq) b. FeO3(s) + HNO3(aq) Fe(NO3)3(aq) + HO(1) c. NH3(g) + O(g) NO(g) + HO(g) d. PC1,(1) + HO(1) H3PO4(aq) +...
-
A common demonstration in chemistry courses involves adding a tiny speck of manganese(IV) oxide to a concentrated hydrogen peroxide (H 2 O 2 ) solution. Hydrogen peroxide decomposes quite...
-
Use the result of Example 28-6 to estimate the X-ray wavelength emitted when a Co (Z = 27) atom jumps from n = 2 to n = 1.
-
Part A Branxton Ltds inventory on 1 April 2019 had a cost of $100 000 and a retail value of $170 000. During April, the companys net purchases cost $216 000 and had a net retail value of $324 000....
-
A growing niche in the restaurant business is gourmet-casual breakfast, lunch, and brunch. Chains in this group include Panera Bread. Suppose that the mean per-person check for breakfast at Panera...
-
What sample size is large enough to assume that the sampling distribution of the proportion is normally distributed?
-
Given the sample data, how can we determine confidence intervals when the standard deviation of population is unknown?
-
In groups of four or five, consider the following information. On 1 July 2019, Stevenson Pty Ltd, a proprietary company with three shareholders, acquired some property by issuing 100 000 shares to...
-
Tascon Corporation sells coffee beans, which are sensitive to price fluctuations. The following inventory information is available for this product at December 31, 2017. Instructions Calculate...
-
Prove the result that the R 2 associated with a restricted least squares estimator is never larger than that associated with the unrestricted least squares estimator. Conclude that imposing...
-
The mobility of a u- ion in aqueous solution is 4.01 x 10-8 m2 S-1 V-1 at 25C. The potential difference between two electrodes placed in the solution is 12.0 V. If the electrodes are 1.00 cm apart,...
-
What fraction of the total current is carried by er when current flows through an aqueous solution of NaCI at 25C?
-
What fraction of the total current is carried by er when current flows through an aqueous solution of NaCI at 25C?
-
D.L. Ko and Company, a manufacturer of quality handmade walnut bowls, has had a steady growth in sales for the past 5 years. However, increased competition has led Mr. Ko, the president, to believe...
-
McCullough Hospital uses a job-order costing system to assign costs to its patients. Its direct materials include a variety of items such as pharmaceutical drugs, heart valves, artificial hips, and...
-
Accounts receivable 37,100 Inventory 68,900 Equipment 350,860 Accumulated depreciation, equipment 159,000 Accounts payable 127,200 Bank loan payable 74,200 Deferred revenue 23,320 Common shares...

Study smarter with the SolutionInn App


