Label the type of bonding for each of the following. a. b.
Question:
Label the type of bonding for each of the following.
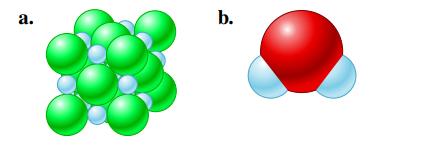
Transcribed Image Text:
a. b.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
Bond refers to the relationship involving two atoms in a chemical or molecule In an ionic molecule p...View the full answer

Answered By

Saqlain Jh
I graduated with a bachelors of commerce degree accounting option and got a second class upper division.am good with numbers, meeting deadlines and working under pressure .I have been an online tutor for the last two years.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
The table shows the type of bonding in a number of elements and compounds. a. Draw a labelled diagram to show metallic bonding. b. Explain why magnesium chloride has a high melting point but bromine...
-
Describe the type of bonding that exists in the F 2 (g) molecule. How does this type of bonding differ from that found in the HF(g) molecule? How is it similar?
-
What is the type of bonding in diamond? Are the properties of diamond commensurate with the nature of the bonding?
-
Write structures for the following bicyclic alkanes: (a) Bicyclo [1.1.0] butane (b) Bicyclo [2.1.0] pentane (c) 2-Chlorobicyclo [3.2.0] heptane (d) 7-Methylbicyclo [2.2.1] heptane
-
Katie and Alan are avid boaters and water skiers. They also enjoy parasailing. This year, they started a new parasailing venture to give rides to patrons. Katie and Alan are both employed full-time...
-
Alton Inc. is working at full production capacity producing 20,000 units of a unique product. Manufacturing costs per unit for the product are Direct materials ............................ $ 9 Direct...
-
What are some of the technological issues that arise through the use of technology in the function of selection?
-
Wallace Corporation summarizes the following information from its weekly payroll records during April. Prepare the two journal entries to record the payment of the payroll and the accrual of its...
-
Protax is a professional tax preparation software for tax accountants. The cost of the software is $7000 per year using "pay as you go" pricing, so on top of the yearly subscription charge, protax...
-
As the Budgeting Officer for Best Fit Ltd, you are required to calculate the budget variances for the financial year FY 202X. The data for the Performance Report is below: Items Sales Cost of sales...
-
Before an electrocardiogram (ECG) is recorded for a cardiac patient, the ECG leads are usually coated with a moist paste containing sodium chloride. Why is sodium chloride applied to the leads?
-
List some characteristic properties that distinguish the metallic elements from the nonmetallic elements.
-
Selected horizontal percentages of a base-year amount from Coastal Ltd.'s income statement are listed here: Assuming that Coastal did not have any non-operating or irregular items, did its profit...
-
The large amount of highly personal data available to employers provides an opportunity to better understand employee behavior. These safety and health data can help uncover ways to support employees...
-
Based in France and operating in 140 countries, LOral is a global corporate giant. It employs more than 89,000 workers and owns brands such as Maybelline, Ralph Lauren, and Lancme among its...
-
You work on a team with four other members of the customer service team at C-Zone, an auto-parts wholesaler. Your company has around 2,000 employees nationally. The company has low job acceptance...
-
A manufacturing company is preparing to implement a company wellness program within its East Coast, Southeastern, and West Coast locations. These locations employ about 1,800 workers in total. The...
-
Imagine that you are working at a medium-sized business as an HR professional. You are faced with the following dilemmas. Decide how you would handle each issue in the short term and long term. What...
-
For a BCC single crystal, would you expect the surface energy for a (100) plane to be greater or less than that for a (110) plane? Why? (Note: You may want to consult the solution to Problem 3.55 at...
-
Write the general quadratic equation y2 - 8y - 4x + 28 = 0 in standard form. Determine the vertex, focus, and directrix of the parabola defined by this equation. Sketch a graph.
-
Normalize the following wave functions: (a) Sin (nx/L) in the range 0
-
Identify which of the following functions are eigen functions of the operator d/dx: (a) D2/dx2, (b) Cos kx, (c) K, (d) Kx, (e) e-ax2. Give the corresponding eigen value where appropriate.
-
Which of the functions in Problem 8.15 are? (a) Also Eigen functions of d2/dx2 and (b) Only Eigen functions of d2/dx2? Give the Eigen values where appropriate.
-
According to the Adapting to Change box, what is the professional is best able to detect the financial irregularities that often accompany fraud in an organization?
-
Sharon Lee recently opened her own basket-weaving studio. She sells finished baskets in addition to the raw materials needed by customers to weave baskets of their own. Sharon has put together a...
-
Here is the income statement for Ivanhoe, Inc. Ivanhoe, Inc. Income Statement For the Year Ended December 31, 2025 Net sales $449,500 Cost of goods sold 211,500 Gross profit 238,000 Expenses...

Study smarter with the SolutionInn App


