Locate the following points in the Bi-Sb phase diagram and indicate the phases present and their relative
Question:
Locate the following points in the Bi-Sb phase diagram and indicate the phases present and their relative amounts:
(a) 60 at% Bi at 250°C;
(b) 30 at% Bi at 500°C; and
(c) 50 at% Bi at 600°C. (See Figure 10-20.)
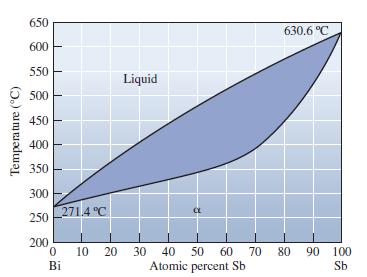
Transcribed Image Text:
Temperature 650 600 550 500 450 400 350 300 250 271.4°C 200 200 0 Bi 10 Liquid CL 20 30 40 50 60 70 Atomic percent Sb 630.6 °C 80 90 100 Sb
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
ANSWER Such questions must be answered using the lever rule There are an endless number of conceivab...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Locate the following points in the Ag-Pd phase diagram and indicate the phases present and their relative amounts: (a) 50 wt% Pd at 1300C; (b) 80 wt% Pd at1425C; and (c) 90 wt% Ag at 1100C....
-
Express the following points in Cartesian coordinates: (a) P(1, 60, 2) (b) Q(2, 90, 4) (c)R(, 45, 210) (d) T(4, /2, /6)
-
Express the following points in cylindrical and spherical coordinates: P(1, 4 , 3)
-
What are the Key Success factors in the Daycare industry? explain
-
Indicate the type of fiscal and monetary policies required to reach point F in the figure for Problem 12 for points C 4 , C 8 , and C 10 .
-
Nautical Marina needs to raise \(\$ 1.0\) million to expand the company. Nautical Marina is considering the issuance of either - \(\$ 1,000,000\) of \(8 \%\) bonds payable, or - 100,000 common shares...
-
Refer to Exercise 12. Compute the value of the test statistic. Exercise 12 A simple random sample of 17 business majors from a certain university had a mean GPA of 2.81 with a standard deviation of...
-
Oakwood Company produces maple bookcases to customer order. It received an order from a customer to produce 5,000 bookcases. The following information is available for the production of the...
-
Using equal amounts of resources, the city of Cliffordland can produce 10 cars of 10 trucks, whereas the city of Econberg can produce 4 cars or 8 trucks. 1. Which country has the absolute advantage...
-
Aunt Tillie has deposited $33,000 today in an account which will earn 10 percent annually. She plans to leave the funds in this account for seven years earning interest. If the goal of this deposit...
-
What is an alloy? In general, what is the difference in the melting behavior of an alloy from that of a pure component?
-
Determine the composition range in which the Tl-Pb alloy at 350C is (a) Fully liquid; (b) Fully solid; and (c) Partly liquid and partly solid. Refer to Figure 10-8(d) for the Tl-Pb phase diagram....
-
Leo Moser studied the effect that two face-to-face panes of glass have on light reflected through the panes. If a ray is unreflected, it has just one path through the glass. If it has one reflection,...
-
Random tunes. Write a program that uses StdAudio to play random tunes. Experiment with keeping in key, assigning high probabilities to whole steps, repetition, and other rules to produce reasonable...
-
The proposed design of a fuel element for a nuclear power reactor consists of 2-in.-diameter cylindrical fuel core surrounded by a 0.25-in.- thick aluminum-alloy cladding. The outside surface of the...
-
(a) Show that the variancecovariance matrix of the disturbances in (9.1) is given by (9.2). (b) Show that the two nonzero block matrices in (9.2) can be written as in (9.3). (c) Show that...
-
A tower packed with 25 m m 25 m m ceramic Raschig rings is to be used for absorbing benzene vapor from a dilute mixture with an inert gas using a wash oil at 300 K 300 K . The viscosity of the oil...
-
Under the assumption that KMSs market share will increase by 0.25% per year (and the investment and financing will be adjusted as described in Problem 13), you project the following depreciation:...
-
Use the data in P13-5 for Prince Company. Assume that the stock price per share is $28 and that dividends in the amount of $3.50 per share were paid during Year 2. Compute the following ratios: ...
-
Explain the buyers position in a typical negotiation for a business. Explain the sellers position. What tips would you offer a buyer about to begin negotiating the purchase of a business?
-
What is the hybridization of the underlined nitrogen atom in each of the following molecules or ions? a. NO + b. N 2 O 3 (O 2 NNO) c. NO 2 - d. N 2
-
Which of the following statement(s) is(are) true? a. The alkali metals are found in the earths crust in the form of pure elements. b. Gallium has one of the highest melting points known for metals....
-
Calculate the solubility of Mg(OH) 2 (K sp = 8.9 10 -12 ) in an aqueous solution buffered at pH = 9.42.
-
One of the four basic types of problems identified by Drucker (2008) is the generic event that is common within the organization and throughout the industry. He goes on to tell us that all but the...
-
Joanne bought a gift for her friend's birthday from an online website. The service provider failed to deliver the gift on the day it had promised. To compensate, the service provider provided coupons...
-
Dr. D is the director of surgery at Union Medical Center. He has long been recognized for his disruptive behavior and outbursts of anger in the OR. Carol, the VP of nursing, makes an appointment to...

Study smarter with the SolutionInn App


