Of the metals listed in Table 22-1, which have the highest and lowest specific heats? Also list
Question:
Of the metals listed in Table 22-1, which have the highest and lowest specific heats? Also list their atomic masses
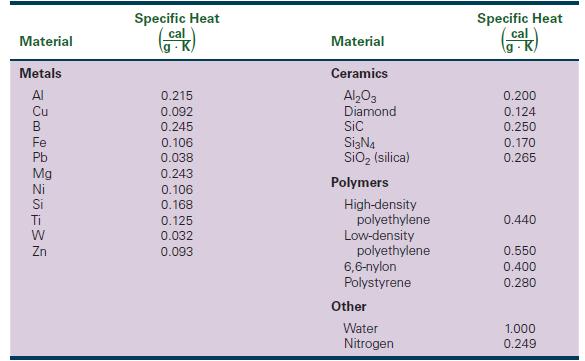
Transcribed Image Text:
Material Metals Al Cu B Fe Pb Mg Ni Si Ti W Zn Specific Heat cal 0.215 0.092 0.245 0.106 0.038 0.243 0.106 0.168 0.125 0.032 0.093 Material Ceramics Al₂O3 Diamond Sic Si3N4 SiO₂ (silica) Polymers High-density polyethylene Low-density polyethylene 6,6-nylon Polystyrene Other Water Nitrogen Specific Heat cal K 0.200 0.124 0.250 0.170 0.265 0.440 0.550 0.400 0.280 1.000 0.249
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
ANSWER From the table we can see that the metal with the highest specific heat is magnesium Mg ...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Of the metals listed in Table 19-1, which are the most and least conductive? Material Superconductors Hg, Nb3Sn YBaCu3O7-x MgB Metals Alkali metals Na K Alkali earth metals Mg Ca Group 3B metals Al...
-
In Figure 20-9(b), what materials have the highest and lowest saturation magnetizations and coercivities? Square loop for computer applications Inductance (a) Soft magnet for electrical applications...
-
For each of the metals listed in the following table, compute the Pilling-Bedworth ratio. Also, on the basis of this value, specify whether you would expect the oxide scale that forms on the surface...
-
Which of the following are valid in a Java file, listed in the order in which they are declared? (Choose two.) A. A package-private class declaration and a public interface declaration B. Two package...
-
In the business world of the 21st Century, will it be possible to make critical marketing decisions without marketing research? Why or why not?
-
(a) Complete the structure of the pain-relieving drug ibuprofen on the basis of the fact that ibuprofen is a carboxylic acid that has the molecular formula C13H18O2, X is an isobutyl group, and Y is...
-
In Exercises 1518, use the given set of points to a. Compute b1. b. Compute the residual standard deviation se. c. Compute the sum of squares for x, (x x)2. d. Compute the standard error of b1, sb....
-
Sam Manuel has been employed on a full-time basis as an electrical engineer for the past three years. Prior to obtaining full-time employment, he was self-employed as an inventor of complex...
-
Geary Company is considering an investment costing $110,000. The investment would return $40,000 per year in each of five years. The investment will require working capital of $30,000. Geary requires...
-
Financial Analyses Provide answers based on both qualitative and quantitative analyses to the following questions: How has Amazon performed leading up to its 20-year-anniversary as a public company?...
-
Calculating Power in Decibels. In an optical communications system or electrical power transmission system, the power or signal often is transferred between several components. The decibel (dB) is a...
-
What is the minimum accelerating voltage required to produce Ka x-rays in nickel?
-
Show that braces do not have to occur in matching pairs inside a string literal.
-
Explain how a ventures sustainable growth rate is related to its financing needs.
-
Describe the main factors that determine a ventures sustainable growth rate. What are the key assumptions in the sustainable growth model?
-
If you were trying to simulate the value of a call option, why would you first simulate the value of the underlying stock rather than simulating the option risk directly?
-
How does Rule 144 restrict the ability of shareholders to liquidate their positions? Why might such restrictions be important to VC investors? Describe conceptually how you would attempt to value a...
-
What is the accounting impact of negative shareholders equity? How can a venture with negative book equity continue to operate and attract capital?
-
Tisdale Incorporated reports the following amount in its December 31, 2018, income Required: 1. Prepare a multiple-step income statement. 2. Explain how analyzing the multiple levels of profitability...
-
In order to get an idea on current buying trends, a real estate agent collects data on 10 recent house sales in the area. Specifically, she notes the number of bedrooms in each house as follows: a....
-
As quality control engineer for your company, you must approve all material shipments from your suppliers. Part of this job involves testing random samples from each delivery and making sure they...
-
When estimating the live load for a new bridge design, you want that estimate to be conservative. In other words, you want to error on the safe side by basing the estimate on the worst possible...
-
Determine the forces acting on the ends of the rope by (1) drawing the free-body diagram, (2) counting unknowns and equations to check determinacy, (3) writing the equilibrium equations, and (4)...
-
Georgeland Cycles makes and sells two models of electric bicycles. The Commuter (a folding model) sells for $2,505.00 and the Tour- X (a fat-tire trail model) sells for $4,505.00. Unit variable costs...
-
How do resistance genes alter the effectiveness of antibiotics that target proteins ?
-
Able Co. received $20,000 from a customer on April 1 for services to be provided partially in April and the rest in May. At the end of April, 11% of the services were complete. What is the amount...

Study smarter with the SolutionInn App


