Sketch the galvanic cells based on the following overall reactions. Show the direction of electron flow, the
Question:
Sketch the galvanic cells based on the following overall reactions. Show the direction of electron flow, the direction of ion migration through the salt bridge, and identify the cathode and anode. Give the overall balanced equation. Assume that all concentrations are 1.0 M and that all partial pressures are 1.0 atm.
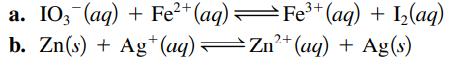
Transcribed Image Text:
2+ a. IO3(aq) + Fe²+ (aq) — Fe³+ (aq) + 1₂(aq) = b. Zn(s) + Ag+ (aq) Zn²+ (aq) + Ag(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
ANSWER a The galvanic cell for the specified overall re...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Sketch the galvanic cells based on the following half-reactions. Calculate Ïo, show the direction of electron flow and the direction of ion migration through the salt bridge, identify the...
-
Sketch the galvanic cells based on the following overall reactions. Calculate o , show the direction of electron flow and the direction of ion migration through the salt bridge, identify the cathode...
-
Assume both electron and hole concentrations in a semiconductor are raised by n above their equilibrium values. Define a net minority carrier lifetime t by R = n/t. give expressions for t in terms of...
-
Which of the following statements about an acquiescence is correct? a. Acquiescences are published only for certain regular decisions of the U.S. Tax Court. b. The IRS does not issue acquiescences to...
-
Robert Shiller of Yale University has suggested a variation on ARMs in which mort-gage interest rates are tied to inflation, not to short-term interest rates. Discuss the pros and cons of this idea...
-
Using EES (or other) software, determine the effect of the amount of air on the adiabatic flame temperature of liquid octane (C8H18). Assume both the air and the octane is initially at 25C. Determine...
-
A long, uninsulated steam line with a diameter of \(100 \mathrm{~mm}\) and a surface emissivity of 0.8 transports steam at \(150^{\circ} \mathrm{C}\) and is exposed to atmospheric air and large...
-
On January 1, the total market value of the Tysseland Company was $60 million. During the year, the company plans to raise and invest $30 million in new projects. The firms present market value...
-
Provide a constructive comparison between the scientific management Human Relations human resource approach as well as the strategic human resource management approach?
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
Calculate G and K at 25 C for the reactions in Exercises 37 and 41. Data in Exercise 37 Sketch the galvanic cells based on the following overall reactions. Show the direction of electron flow, and...
-
Which of the following statements concerning corrosion is(are) true? For the false statements, correct them. a. Corrosion is an example of an electrolytic process. b. Corrosion of steel involves the...
-
Identify the best strategies for communicating change.
-
Rinaldo informs you that the first transaction that you will have to record is a cash payment transaction. On May 1, 2017, Check No. 4041 was issued to 'Office Supplies Warehouse' for the cash...
-
A company has fixed costs of $10,000 and it profits $42,250 off of 2,750 items. Find the linear profit function, P(x) P(x) =
-
Short strangle options trading strategy consists of writing a call and a put option at different exercise prices. Construct a profit/loss diagram for this option strategy given the following...
-
identify an innovation or change in the Financial Services industry, which has changed the way the industry does business in this area of financial services
-
Look at the Wag! website and determine the 4Ps of this service. Do they work well together? Why or why not
-
Draw the converse of each of the graphs in Exercises 7-9 in Section 10.1.
-
Compare and contrast licensing and subcontracting.
-
If an electron passes through an electrical potential difference of 1 V, it has an energy of 1 electron-volt. What potential difference must it pass through in order to have a wavelength of 0.300 nm?
-
Calculate the longest and the shortest wavelength observed in the Balmer series.
-
X-rays can be generated by accelerating electrons in a vacuum and letting them impact on atoms in a metal surface. If the 1250. eV kinetic energy of the electrons is completely converted to the...
-
1. What year are you preparing your students for? 1973? 1995? Provide evidence that your school's curriculum and the program you use are preparing your students for 2030? Are you even preparing them...
-
2 Classify each type of numerical data as discrete (D) or continuous (C). a number of homes with solar power b patients' body temperatures c sizes of the crowds at rock concerts d star ratings of...
-
What role does the use of either mimesis or abstraction play in each example? (Keep in mind that the concepts of mimesis and abstraction in the creative arts are defined and explored in the document...

Study smarter with the SolutionInn App


