Specify which of the following are oxidationreduction reactions, and identify the oxidizing agent, the reducing agent, the
Question:
Specify which of the following are oxidation–reduction reactions, and identify the oxidizing agent, the reducing agent, the substance being oxidized, and the substance being reduced.
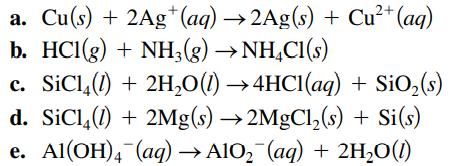
Transcribed Image Text:
2+ a. Cu(s) + 2Ag+ (aq) →2Ag(s) + Cu²+ (aq) b. HCl(g) + NH3(g) →NH4Cl(s) c. SiC¹4(1) + 2H₂O(l) →4HCl(aq) + SiO₂ (s) d. SiC14 (1) + 2Mg(s) →2MgCl₂(s) + Si(s) e. Al(OH)4 (aq) →AlO₂ (aq) + 2H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a This is an oxidationreduction reaction Oxidizing ...View the full answer

Answered By

Hande Dereli
Enthusiastic tutor, skilled in ACT and SAT tutoring. Raised one student's score on the SATs from 1100 combined to 1400. Graduated with a 3.9 GPA from Davidson College and led a popular peer tutoring group for three years. Scored in the top 0.06% in the nation on the SATs. The real reason I'm the one to help you nail the test? Results. Clients invariably praise my ability to listen and communicate in a low-stress, fun way. I think it's that great interaction that lets me raise retest SAT scores an average of 300 points.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Specify which of the following elements you would expect to have the greatest electron affinity and which would have the least: He, K, Co, S, Cl.
-
Identify with a which of the following are subdivisions of owners equity. a. Vehicles _____ b. J. Penny, Capital _____ c. Accounts Payable _____ d. J. Penny, Withdrawals _____ e. Accounts Receivable...
-
Identify with a which of the following are subdivisions of owners equity. a. Land b. M. Kaminsky, Capital c. Accounts Receivable d. M. Kaminsky, Withdrawals _____ e. Accounts Payable _____ f. Rent...
-
What conditions must be met for an award to qualify for an exclusion under Sec. 74?
-
Discuss the role of perception in the stress response. You may find it useful to review the perception portion of Chapter 5 and discuss its implications for understanding the stress response.
-
Using information from the preceding question, suppose Copies R Us sold the machine for $5,000. What is the result of this disposal transaction? a. Gain of $2,000 b. Loss of $2,000 C. Gain of $3,000...
-
Please discuss the video "Fog of War" and proportionality in war. What was McNamara saying about proportionality during World War II? How do you think this impacted the decision making processes...
-
A building supply company is manufacturing residential doors. Because demand has been increasing for their products, the company is trying to evaluate whether to add a second production line. For...
-
Krazy Kars Pty Ltd (Krazy Kars) equips and sells customised off-road vehicles to operators in the tourist industry. The directors are Carlos, Possum and Michele. According to its constitution,...
-
A 30.0-mL sample of an unknown strong base is neutralized after the addition of 12.0 mL of a 0.150 M HNO 3 solution. If the unknown base concentration is 0.0300 M, give some possible identities for...
-
A 25.00-mL sample of hydrochloric acid solution requires 24.16 mL of 0.106 M sodium hydroxide for complete neutralization. What is the concentration of the original hydrochloric acid solution?
-
Ms. Schmidt didnt request an extension of time to file a 2022 income tax return and didnt mail the completed return to the IRS until August 8, 2023. Ms. Schmidt enclosed a check for $2,380, the...
-
Under what circumstances is using powerless speech advantageous?
-
What are some ways you might undersell yourself on rsums?
-
Which forms of language are characterized as powerless?
-
Why should you use action words on your rsums?
-
As an aspect of credibility, how is experience different from expertise?
-
Sarasota Company has a credit balance of $2,200 in Allowance for Doubtful Accounts before adjustment. The estimated uncollectibles under the percentage- of-receivables basis is $5,100. Prepare the...
-
For the next several days, take notes on your listening performance during at least a half-dozen situations in class, during social activities, and at work, if applicable. Referring to the traits of...
-
The centre of the EPR spectrum of atomic deuterium lies at 330.02 mT in a spectrometer operating at 9.2482 GHz. What is the g-value of the electron in the atom?
-
A radical containing three equivalent protons shows a four-line spectrum with an intensity distribution 1:3:3:1. The lines occur at 331.4 mT, 333.6 mT, 335.8 mT, and 338.0 mT. What is the hyperfine...
-
A radical containing three in equivalent protons with hyperfine constants 2.11 mT, 2.87 m'T and 2.89 mT gives a spectrum centred on 332.8 mT. At what fields do the hyperfine lines occur and what are...
-
The following are extracted from the Cash Book of Jungle Trading and its Bank Statement (received from ABC Bank) for the month of April 2020: Cash Book April 2020 RM April 2020 1 Bal b/d 1,500 9...
-
1. Briefly describe FIVE (5) differences between qualitative data and quantitative data. 2. The literature review is an integral part of the research process and make a valuable contribution to...
-
Writing a detailed written assessment that challenges the traditional legal regimes governing the use of military force, addressing key jus ad bellum and jus in bello legal concerns along with their...

Study smarter with the SolutionInn App


