The active ingredient of aspirin tablets is acetylsalicylic acid, which has a density of 1.4 g/cm 3
Question:
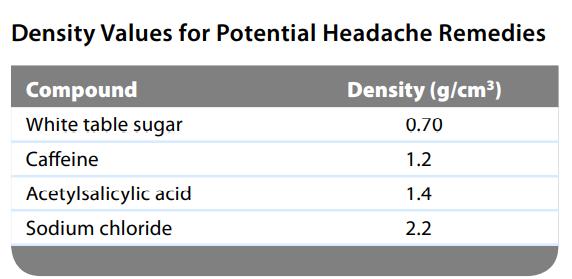
The active ingredient of aspirin tablets is acetylsalicylic acid, which has a density of 1.4 g/cm3. In a lab class, a student used paper chromatography to isolate another common ingredient of headache remedies. The isolated sample had a mass of 0.384 g and a volume of 0.32 cm3. Given the data in the following table, what was the other ingredient in the headache remedy?
Transcribed Image Text:
Density Values for Potential Headache Remedies Density (g/cm³) Compound White table sugar Caffeine Acetylsalicylic acid Sodium chloride 0.70 1.2 1.4 2.2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
Other active ingredient found in a headache remedy is iontophylates ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
The active ingredient of the insect repellent Off is N, N-diethyl-m-toluamide, m-CH3C6H4CON (CH2CH3)2. Outline a synthesis of this compound starting with 3-methylbenzoic acid (m-toluic acid).
-
Repeat Problem 69 for cobalt, which has a density of 8.9 g/cm3, a molecular mass of 58.9 g/mol, and a saturation magnetization given by 0Ms = 1.79 T.
-
Castor oil, which has a density 0.96 x 103 kg/m 3 at room temperature, is forced through a pipe of circular cross section by a pump that maintains a gauge pressure of 950 pa the pipe has a diameter...
-
General-equilibrium effects with labor complementarity. Consider an economy comprised of 100 cities. Each city initially contains 1 million each of high school dropouts, high school graduates,...
-
Lynette, a famous basketball player, is considering the possibility of transferring the sole right to use her name to promote basketball shoes produced and sold by the NIK Corporation. NIK will pay...
-
Let (x) = -3x + 4 and g(x) = -x 2 + 4x + 1. Find each of the following. Simplify if necessary. g (10)
-
Fill in the Blank. __________ principle is not applicable in nonlinear analysis.
-
Proration of overhead the Ride-On-Water Company (ROW) produces a line of non-motorize boats. ROW uses a normal costing system and allocates manufacturing overhead using direct manufacturing labor...
-
The following Information has been provided for the City of Elizabeth for its fiscal year ended June 30. The Information provided relates to financial Information reported on the city's statement of...
-
In the Chapter 3 Data Analytics Case, you applied Tableau to examine a data set and create calculations to compare two companies profitability. For the case in this chapter, you continue in your role...
-
Lipitor, a pharmaceutical drug that has been shown to lower bad cholesterol levels while raising good cholesterol levels in patients taking the drug, had over $3 billion in sales in 2015. Assuming...
-
A 194-g sample of caffeine (C 8 H 10 N 4 O 2 ) contains 6.02 10 23 molecules of caffeine. If a typical 10-hour energy drink contains 422 mg of caffeine, how many molecules of caffeine are present in...
-
Johnson Corporation purchased all of the outstanding common stock of Smith Corporation for $11,000,000 in cash. The book value of Smith's net assets (assets minus liabilities) was $7,800,000. The...
-
Q1. Many of the characters show intense resentment and bitterness toward people they must interact with every day. What creates these emotions? Conflicts easily and often result. What do you think...
-
Answer the below Question and also explain the types of cost discuss in this Question ? Marshal's JPMorgan Chase credit card has a 15 percent interest rate and a rewards program that gives him one...
-
Question Gross Rent Multiplier uses gross income and the formula is: Value/Gross Annual Income = Gross Rent Multiplier. A property generates gross rents of $40,000 yearly. It recently sold for...
-
The table below reports real GDP per capita for Germany and the United States for 2008 and 2018. Country Germany the United States Per capita real GDP 2008 Per capita real GDP 2018 $42,102 $49,319...
-
What does this map tell us about the intricate Facebook relationships and the impact or influence of globalization? a. Does this mean there is no social media in places like China and Russia? b. Is...
-
Melinda Stoffers owns and operates ABC Print Co. During February, ABC Print Co. incurred the following costs in acquiring two printing presses. One printing press was new, and the other was bought...
-
You are a Loan Officer with an Investment Bank. Today you need to set your lending parameters. They are: LTV: 55% 10 Year T-Bill: TBD Rate Markup: 300 Basis Points Term: 30 Years Amortization: 30...
-
What is the most probable point (not radius) at which a 2p electron will be found in the hydrogen atom?
-
Explicit expressions for hydrogenic orbitals are given in Tables 10.1 and 9.3. (a) Verify both that the 3px orbital is normalized (to I) and that 3px and 3dxy are mutually orthogonal. (b) Determine...
-
Show that l, and 12 both commute with the Hamiltonian for a hydrogen atom. What is the significance of this result?
-
The amount spent per month by employees for parking at a municipal lot is normally distributed with a mean of $540 and a standard deviation of $95. (Round the final answers to 2 decimal places.) a....
-
Let A = 3 Find B = sin(A) Find C = A Find D=C2 Find ECC Find F = C.*C % Your script goes here.
-
Determine the highest real root of f(x) = -3x - 4x + 12 2 a) by graphing it in MATLAB (sketch your plot and give an approximate value) b) using 3 iterations (by hand) of Newton-Raphson. Make a table...

Study smarter with the SolutionInn App


