The barometer below shows the level of mercury at a given atmospheric pressure. Fill all the other
Question:
The barometer below shows the level of mercury at a given atmospheric pressure. Fill all the other barometers with mercury for that same atmospheric pressure. Explain your answer.
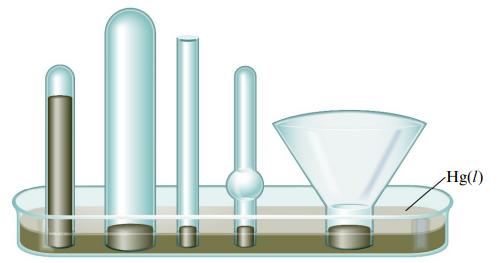
Transcribed Image Text:
M 1 Hg(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
My explanation is that the atmospheric pressure a...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
A barometer to measure absolute pressure shows a mercury column height of 725 mm. The temperature is such that the density of the mercury is 13550 kg/m3. Find the ambient pressure.
-
A barometer to measure absolute pressure shows a mercury column height of 725 mm. The temperature is such that the density of the mercury is 13 550 kg/m3. Find the ambient pressure.
-
Atmospheric pressure P at an altitude a is given by P = P0e-0.00005a, Where P0 is the pressure at sea level, approximately 14.7 lb/in2 (pounds per square inch). Explain how a barometer, or some...
-
The unadjusted trial balance of Mesa Inc., at the company's year end of December 31 follows: Additional information and adjustment data: 1. The 12-month insurance policy was purchased and was...
-
Mr. Bahner was hosting a dinner for friends at a restaurant in the Bayshore Inn, Vancouver. The group ordered one bottle of wine and, at 11:30, the waiter asked if they would like another, to which...
-
In view, of the provisions of the Chartered Accountants Act, 1949 state and justify your views on the following cases (a) Whether you would accept offer of appointment as auditor of a firm in which...
-
How to draft a pretrial memorandum?
-
A 2.20.kg hoop 1.20 m in diameter is rolling to the right without slipping on a horizontal floor at a steady 3.00 rad/s. (a) How fast is its center moving? (b) What is the total kinetic energy of the...
-
assume you are explaining to a customer how to identify ammunition cartridges. Explain the purpose and use of a headstamp. What are the elements found on cartridges? Provide some examples of the...
-
Create the following 6 financial analysis reports for Casey Corporation and provide an analysis as requested: a. Income Statement (Horizontal) report with a run date of 12/31/. Provide a narrative...
-
A diagram in a chemistry book shows a magnified view of a flask of air as follows: What do you suppose is between the dots (the dots represent air molecules)? a. Air b. Dust c. Pollutants d. Oxygen...
-
What are Boyles law, Charless law, and Avogadros law? What plots do you make to show a linear relationship for each law?
-
What is the normal due date for the tax return of calendar-year taxpayers? What happens to the due date if it falls on a Saturday, Sunday, or holiday?
-
The following transactions relate to a dairy, converting milk to cheese, for the month of May. Prepare ledger accounts which record the transactions. 1 May Bought 600 drums of milk from supplier on...
-
Alternatives 1, 2, and 3 have lives of 3, 4, and 6 years, respectively. Their net cash flow (NCF) and salvage value (SV) profiles are as follows: Additional explanation is necessary: the NCF profile...
-
Write down five ways in which budgets appear to benefit an organisation. Then read the next section. How does your list compare with the text? Have you identified benefits additional to those...
-
Irving Fisher developed the clearest exposition of the classical quantity theory of money. Go to https://en.wikipedia.org/wiki/Irving_Fisher and write a onepage summary of his life and contributions.
-
Consider the following cash flows and interest rates: a. Determine the future worth of this series of cash flows. b. Determine the present worth of this series of cash flows. c. Determine a 3-year...
-
Refer to the financial statement information of Under Armour reprinted at the back of the book. Required 1. Using the format in Example 13-5, prepare common-size comparative income statements for...
-
The trade-off theory relies on the threat of financial distress. But why should a public corporation ever have to land in financial distress? According to the theory, the firm should operate at the...
-
Do you expect cyclohexene to adopt a chair conformation? Why or why not? Explain. Cyclohexene
-
For each pair of compounds below, determine whether they are identical compounds, constitutional isomers, stereoisomers, or different conformations of the same compound: (a) (b) (c) (d) (e) (f) (g)...
-
Predict the major product(s) for each of the following reactions: 1) Hg(OAC)2, -0 2) NABH, ? - O, NaOH, cold Br2 H20 Pt
-
Select a company you are familiar with and detail one short-term and one longer-term strategy, then discuss how the role of human resource development can provide valuable advice and counsel in the...
-
Sunspot Beverages, Limited, of Fiji uses the weighted-average method of process costing. It makes blended tropical fruit drinks in two stages. Fruit juices are extracted from fresh fruits and then...
-
How does TPM intersect with Lean Manufacturing principles? Discuss the synergies between TPM and Lean practices, such as continuous improvement (Kaizen) and value stream mapping, in reducing waste...

Study smarter with the SolutionInn App


