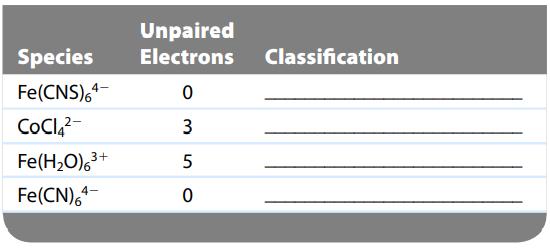
The following table indicates the number of unpaired electrons in the crystal field diagrams for some complexes.
Question:
The following table indicates the number of unpaired electrons in the crystal field diagrams for some complexes. Complete the table by classifying each species as weak field, strong field, or insufficient information.
Transcribed Image Text:
Unpaired Species Electrons Classification Fe(CNS)64 0 CoCl₂²- 3 5 0 Fe(H₂O)63+ Fe(CN)64 4-
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
Blood sample may be collected over 24 hour at the vo...View the full answer

Answered By

Dudhat Vaidehi
I tutored mostly elementary school students privately after school and during the summer. We met in their homes or at the public library. I charged an hourly fee, and I provided any necessary materials.
Having taught in special education in two local schools for many years meant that I had contact with a lot of parents of special needs students. I never had to advertise — word of mouth was how most folks knew of me. At one point I did have a website, but didn't utilize it much. I stayed very busy, especially in the summers, and always had a full schedule. I typically met with each student's teacher in order to get an idea of what the focus of my instruction/remediation should be. Becoming familiar with the student's learning style(s) was also very helpful. Often parents would share records and test results with me. After each tutoring session, I documented the student’s progress and gave parents written updates, as well as phone calls or emails as needed.
While my students and I certainly utilized technology and the internet often during our sessions, I never tutored online or for any tutoring company, so am not familiar with the curriculums or methods used in those settings.
Tutoring one on one was very enjoyable and rewarding. My students and I had fun, and grew quite fond of one another. The extra income was a bonus. I had to retire from tutoring due to a physically handicapping disease, and miss my students very much.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
The following table indicates the number of constitutional isomers with molecular formula C 7 H 16 . Draw each of the isomers, making sure not to draw the same compound twice. NUMBER OF...
-
The following table indicates the percentage of residential electricity consumption in the United States, in a recent year organized by type of use Type of Appliance __________Percentage Cooking...
-
The following table indicates the customers buying behavior for a given product Produced by ABC Company with two brands. Currently, the market share for Brand A is 27.5%, Brand B is 37.5% and...
-
First United Bank Inc. is evaluating three capital investment projects by using the net present value method. Relevant data related to the projects are summarized as follows: Instructions 1. Assuming...
-
"Changes in disposable income lead to movements along the consumption function; changes in wealth or other factors lead to a shift of the consumption function." Explain this statement with an...
-
Duncan Industries completed these transactions during July 2020. The terms of all credit sales are 2/10, n/30. Required 1. Set up accounts receivable subledger accounts for Kelly Grody, Karen Harden,...
-
The A-36 steel bars are pin connected at \(B\). If each has a square cross section, determine the vertical displacement of \(B\). 800 lb A B D 8 ft 4 ft 10 ft 2 in. H I2 I 2 in.
-
A U.S.-based multinational company has two subsidiaries, one in Mexico (local currency, Mexican peso, MP) and one in Japan (local currency, yen, ¥). Forecasts of business operations indicate the...
-
Hint(s) Check My Work Cox Electric makes electronic components and has estimated the following for a new design of one of its products: Fixed Cost $9,000 == Material cost per unit = $0.15 Labor cost...
-
Use the attached "words.txt" file to store the words along with their ranks in an AVL tree. Then provide the user the option to search for any word. If the word exists the program displays the rank...
-
Which of the following ions is(are) expected to form colored octahedral aqueous complex ions? a. Zn 2+ b. Cu 2+ c. Mn 3+ d. Ti 4+
-
In which of the following is(are) the electron configuration(s) correct for the species indicated? a. Cu [Ar]4s 2 3d 9 b. Fe 3+ [Ar]3d 5 c. Co [Ar]4s 2 3d 7 d. La [Ar]6s 2 4f 1 e. Pt 2+ [Xe]4f 14 5d 8
-
In December 2018, AB Concrete (AB) discovered that Amber Clark, ABs former volunteer bookkeeper, had been writing company checks made payable to herself and her husband, Kai Clark. The Clarks...
-
Examine, analyze, and compare the activity of the IKEA real estate brand on various media platforms such as Facebook, Instagram, Twitter or Pinterest. Advertisements videos and pictures are posted on...
-
What was the process to prepare a speech? What challenges did you have when preparing your speech? How did you overcome them? What challenges did you have recording your speech? How did you overcome...
-
1. You are an instructor who is having difficulty with students who do not want to work on a group project. Students complain others are not working equally or that they do not like certain people....
-
Does the plot of the opera reflect the ideals of the Romantic period? Why?
-
2. The functions f(x) = x sin x, f2(x) = x cos x, f(x) = sin x, f(x) = cos x span a 4-dimensional subspace V of the vector space F(R). Let T: V F(R) be linear transformation defined by (Tf)(x) = f(x...
-
At what frequency f does a sound wave in air have a wavelength of 15 cm, about half the diameter of the human head? Some methods of localization work well only for frequencies below f, while others...
-
Describe the general ways that the revised Form 990, applicable for tax year 2008 and beyond, is different from previous versions.
-
Which of the other six D aldohexoses yield optically active aldaric acids on oxidation, and which yield optically inactive (meso) aldaric acids?
-
What product(s) would you expect from Kiliani-Fischer reaction of D-ribose?
-
What aldopentose would give a mixture of L-gulose and L-idose on Kiliani-Fischer chain extension?
-
Complete the Price, Advertising, and Profitability chart. a . ?Notice that the series for $ 2 5 , 0 0 0 , 0 0 0 ?is already on the chart. b . ?Add the series for advertising budgets $ 5 0 , 0 0 0 , 0...
-
Solve for the missing information designated by "?" in the following table. (Use 365 days in a year. Round the inventory turnover ratio to one decimal place before computing days to sell. Round days...
-
Grove Media plans to acquire production equipment for $800,000 that will be depreciated for tax purposes as follows: year 1, $320,000; year 2, $180,000; and in each of years 3 through 5, $100,000 per...

Study smarter with the SolutionInn App


