The rate of the reaction depends only on the concentration of nitrogen dioxide below 225 C.
Question:
The rate of the reaction
![]()
depends only on the concentration of nitrogen dioxide below 225οC. At a temperature below 225οC, the following data were collected:
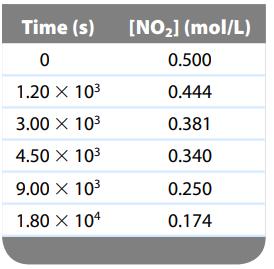
Determine the rate law, the integrated rate law, and the value of the rate constant. Calculate [NO2] at 2.70 × 104 s after the start of the reaction.
Transcribed Image Text:
NO₂(g) + CO(g) NO(g) + CO₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
Solution a The rate law is rate k on time t b The integ...View the full answer

Answered By

James Warinda
Hi! I’m James Otieno and I'm an experienced professional online tutor with countless hours of success in tutoring many subjects in different disciplines. Specifically, I have handled general management and general business as a tutor in Chegg, Help in Homework and Trans tutor accounts.
I believe that my experience has made me the perfect tutor for students of all ages, so I'm confident I can help you too with finding the solution to your problems. In addition, my approach is compatible with most educational methods and philosophies which means it will be easy for you to find a way in which we can work on things together. In addition, my long experience in the educational field has allowed me to develop a unique approach that is both productive and enjoyable.
I have tutored in course hero for quite some time and was among the top tutors awarded having high helpful rates and reviews. In addition, I have also been lucky enough to be nominated a finalist for the 2nd annual course hero award and the best tutor of the month in may 2022.
I will make sure that any student of yours will have an amazing time at learning with me, because I really care about helping people achieve their goals so if you don't have any worries or concerns whatsoever you should place your trust on me and let me help you get every single thing that you're looking for and more.
In my experience, I have observed that students tend to reach their potential in academics very easily when they are tutored by someone who is extremely dedicated to their academic career not just as a businessman but as a human being in general.
I have successfully tutored many students from different grades and from all sorts of backgrounds, so I'm confident I can help anyone find the solution to their problems and achieve
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
The rate of the reaction NO2(g) + CO(g) NO(g) + CO2(g) depends only on the concentration of nitrogen dioxide at temperatures below 225oC. At a temperature below 225oC, the following data were...
-
The rate of the reaction O(g) + NO2(g) NO(g) + O2(g) was studied at a certain temperature. This reaction is one step of the nitric oxide catalyzed destruction of ozone in the upper atmosphere. a. In...
-
The rate of the reaction CH3COOC2H5(aq) + OH (aq) CH3COO(aq) + C2H5OH(aq) Was measured at several temperatures, and the following data were collected: Temperature (oC) k (M-1 s-1)...
-
Write Python code that prompts the user to enter his or her age and assigns the users input to an integer variable named age.
-
What do you believe will be the strongest motivations behind the purchase of an electric car? Do you think the Ontario incentive program is attractive? Do you think this rebate program will cause a...
-
Divide into groups and, under the guidance of a group leader, compare and contrast the credit policies and methods of monitoring cash collections appropriate for the following types of businesses: 1....
-
Outline the overall approach to an internal operational audit. How is this approach similar to other types of audit activities?
-
Luce & Morgan, a law firm in downtown Jefferson City, is considering opening a legal clinic for middle-and low-income clients. The clinic would bill at a rate of $18 per hour. It would employ law...
-
You are the CFO at ABC Inc., a public company listed in Toronto Stock Exchange (TSX). Its basic business is high-temperature treatment services for contaminated soil. It is now December 9 th , 2022,...
-
In Example 1.4 we looked to see whether the competent- face method could be used to predict the results of Senate races. We found this method worked in 23 out of 32 races. When testing to see if the...
-
Hydrogen reacts explosively with oxygen. However, a mixture of H 2 and O 2 can exist indefinitely at room temperature. Explain why H 2 and O 2 do not react under these conditions.
-
The type of rate law for a reaction, either the differential rate law or the integrated rate law, is usually determined by which data is easiest to collect. Explain.
-
How should a firm with self-sufficient production facilities in several ASEAN countries respond to the creation of a single market? What are the constraints on its ability to respond in a manner that...
-
IceKreme Inc. makes ice cream machines for sale to ice cream parlours. The following events occurred between April 1 and June 30 of the current year: April 10 Received an order from Peter's...
-
Vulcan Company's contribution format income statement for June is as follows: Vulcan Company Income Statement For the Month Ended June 30 es Sales Variable expenses Contribution margin Fixed expenses...
-
Sun Sound Ear Bling Headphones Headphones Sales price $29.20 $45.60 Variable cost of goods sold (16.40) (25.50) Manufacturing margin $12.80 $20.10 Variable selling and administrative expenses (5.80)...
-
The following information is available for the assets of Saman Ltd. at December 31, Year 5: Tangible assets (net) Recognized intangible assets (net) Internally developed patent Goodwill Total...
-
Jorgansen Lighting, Incorporated, manufactures heavy-duty street lighting systems for municipalities. The company use variable costing for internal management reports and absorption costing for...
-
(a) Find the magnitude of the angular momentum L for an electron with n = 2 and = 1 in terms of . (b) What are the allowed values for Lz? (c) What are the angles between the positive z -axis and L...
-
Flicker, Inc., a closely held corporation, acquired a passive activity this year. Gross income from operations of the activity was $160,000. Operating expenses, not including depreciation, were...
-
Draw all constitutionally isomeric ethers with molecular formula C 4 H 10 O. Provide a common name and a systematic name for each isomer.
-
Starting with cyclohexene and using any other reagents of your choice, show how you would prepare each of the following compounds. a. b. c. OH OMe
-
When 1, 4-dioxane is heated in the presence of HI, compound A is obtained: a. Draw the structure of compound A. b. If one mole of dioxane is used, how many moles of compound A are formed? c. Show a...
-
Suppose you bought a new home for $210,000 using a 30-year mortgage with monthly payments of $1,218.841. The annual interest rate of the mortgage is 5.7%. After the first 3 years (36 monthly...
-
Rather than use the hypothetical information above, determine from available news sources if the Fed is increasing, holding steady, or decreasing interest rates. Similarly, is the Federal Government...
-
For a two-sided matching problem, we say that matching is Pareto efficient, if there is no matching ', such that and '(x) R (x) for all x WUF, '(x) Px (x) for some x WUF. Show that stability implies...

Study smarter with the SolutionInn App


