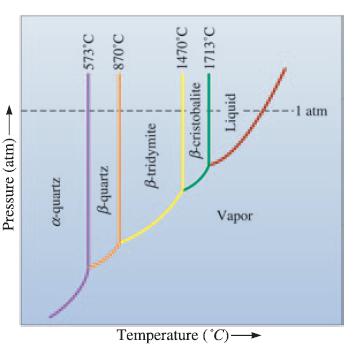
The unary phase diagram for SiO 2 is shown in Figure 10-18. Locate the triple point where
Question:
The unary phase diagram for SiO2 is shown in Figure 10-18. Locate the triple point where solid, liquid, and vapor coexist and give the temperature and the type of solid present. What do the other “triple” points indicate?
Transcribed Image Text:
Temperature (°C)- Pressure (atm)- d-quartz B-quartz Vapor B-tridymite I B-cristobalite Liquid -1 atm 573°C 870°C 1470°C 1713°C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
ANWER Below is seen the unary diagram for SiO2 The unary phase diagram is a particul...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
The phase diagram for water is shown in FIGURE 17-37. (a) What is the temperature T1 on the phase diagram? (b) What is the temperature T2 on the phase diagram? (c) What happens to the...
-
Figure 10-19 shows the unary phase diagram for carbon. Based on this diagram, under what conditions can carbon in the form of graphite be converted into diamond? Pressure (kbars) 700 600 500 400 300...
-
The block diagram of a system is shown in Figure E2.24. Determine the transfer function T(s) = Y(s)/R(s). 10 RIS) FIGURE E2.24 Multiloop feedback system.
-
You are attempting to arrive at what the monthly rate of change that has recently occurred in the real estate market. In doing some market research, you find two properties that have sold twice...
-
Using an ISLM diagram, show the effect of an expansionary fiscal policy on the aggregate demand curve.
-
Two blocks of masses 1.0 kg and 2.0 kg, respectively, are pushed by a constant applied force Facross a horizontal frictionless table with constant acceleration such that the blocks remain in contact...
-
How do you graphically illustrate the System Operations Model?
-
Dieker Container Company is suffering declining sales of its principal product, nonbiodegradeable plastic cartons. The president, Edward Mohling, instructs his controller, Betty Fetters, to lengthen...
-
Answer the questions in the picture below using all listed information 15. Ugh Kinetic Elastic 200 ZE 2 kg 1 m 2 kg 0.5 A 2 kg mass is pushed 0.5 m into a spring with spring constant 200 N/m on a...
-
The managers of United Medtronics are evaluating the following four projects for the coming budget period. The firm's corporate cost of capital is 14%. A. What is the firm's optimal capital budget?...
-
Is copolymer formation similar to solidsolution formation?
-
What is the ABS copolymer? State some of the applications of this material.
-
If you were a native of Brazil and advising American business representatives on what to do when talking with Brazilian business partners, what would you tell the Americans about Brazilian culture?
-
The following are transactions related to property, plant and equipment of Tulsi Sdn Bhd occurred during 2023: Jan 2 June 30 Oct 29 Exchanged old equipment that cost RM69,000 and accumulated...
-
Jordan Corporation began operations on March 1, 2023. The following data is available for March 31: Accounts payable Accounts receivable Cash Contributed capital Expenses, excluding income taxes...
-
You have just been hired by FAB Corporation, the manufacturer of a revolutionary new garage door opening device. The president asked you to review the company's costing system and "do what you can to...
-
On December 31, 2024, the end of the fiscal year, California Microtech Corporation held its semiconductor business for sale at year- end. The estimated fair value of the segment's assets, less costs...
-
How does toyota use the principle of scientific management of this two (science not rule of thumb) and (division of works and responsibilities), explain how they apply both science not rule of thumb...
-
An alkyl halide reacts with an alkoxide ion to form a compound whose 1H NMR spectrum is shown here. Identify the alkyl halide and the alkoxide ion. 0 3 4 6 7 8 9 0
-
How has the too-big-to-fail policy been limited in the FDICIA legislation? How might limiting the too-big-to-fail policy help reduce the risk of a future banking crisis?
-
What are transuranium elements and how are they synthesized?
-
What is a nuclear transformation? How do you balance nuclear transformation reactions? Particle accelerators are used to perform nuclear transformations. What is a particle accelerator?
-
Do radiotracers generally have long or short half-lives? Explain.
-
(a) What is the kinetic energy in joules of a 1140-kg automobile traveling at 93 km/h? (b) How much work would have to be done to bring a 1140-kg automobile traveling at 93 km/h to a stop? 4. How...
-
A charging RC circuit controls the intermittent windshield wipers in a car. The emf is 12.0 V. The wipers are triggered when the voltage across the 120F capacitor reaches 12.0 V; then the capacitor...
-
A block of mass 2 kg is attached to a spring with a spring constant of 100 N/m. The block is released from rest and moves along a rough horizontal surface with a coefficient of friction of 0.2. How...

Study smarter with the SolutionInn App


