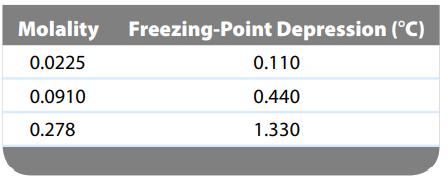
Use the following data for three aqueous solutions of CaCl 2 to calculate the apparent value of
Question:
Use the following data for three aqueous solutions of CaCl2 to calculate the apparent value of the van’t Hoff factor.
Transcribed Image Text:
Molality Freezing-Point Depression (°C) 0.0225 0.0910 0.278 0.110 0.440 1.330
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)

Answered By

Carly Cimino
As a tutor, my focus is to help communicate and break down difficult concepts in a way that allows students greater accessibility and comprehension to their course material. I love helping others develop a sense of personal confidence and curiosity, and I'm looking forward to the chance to interact and work with you professionally and better your academic grades.
4.30+
12+ Reviews
21+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Use the following data for parts (a) through (f). a. Determine the equation of the least squares regression line to predict y by x. b. Using the x values, solve for the predicted values of y and the...
-
Use the following data for parts (a) through (g). a. Determine the equation of the simple regression line to predict y from x. b. Using the x values, solve for the predicted values of y and the...
-
Use the following data for a firm's output at various levels of employment (L) to calculate: a) its marginal physical product of labor (MPPL) schedule; (b) its (MPPL/MRCL) schedule, given a fixed...
-
Contact local employers and ask for copies of their employee handbooks. If none are available, research parent companies of local employers online to see if their employee handbooks are available....
-
What are core and augmented services? How do marketers increase market share with augmented services?
-
Ectocarpene is a volatile, sperm cell-attracting material released by the eggs of the seaweed Ectocarpus siliculosus. Its constitution is
-
The radial component of velocity of water leaving the centrifugal pump sketched in Fig. P12.7 is \(45 \mathrm{ft} / \mathrm{s}\). The magnitude of the absolute velocity at the pump exit is \(90...
-
Beer sales at the Shapiro One-Stop Store are analyzed using temperature and number of people (age 21 or over) on the street as independent variables. A random sample of 20 days is selected, and the...
-
Input is the force f and output is the velocity v2. Find the transfer function in terms of Y(s)/U(s) C m2 m1 V2
-
A rifle is used to shoot twice at a target, using identical cartridges. The first time, the rifle is aimed parallel to the ground and directly at the center of the bull's-eye. The bullet strikes the...
-
Consider the following solutions: 0.010 m Na 3 PO 4 in water 0.020 m CaBr 2 in water 0.020 m KCl in water 0.020 m HF in water (HF is a weak acid.) a. Assuming complete dissociation of the soluble...
-
How would you prepare 1.0 L of an aqueous solution of sodium chloride having an osmotic pressure of 15 atm at 22 C? Assume sodium chloride exists as Na + and Cl - ions in solution.
-
A political poll based on a random sample of 1000 likely voters classified them by sex and asked them if they planned to vote for Candidate A or Candidate B in the upcoming election. Results are...
-
identify which are media and which are information assets: Traverse Accounting Software provides the following applications: General Ledger Accounts Payable Accounts Receivable Payroll (Employee...
-
You are required to find and analyse a personally interesting case study of a successful entrepreneurial for-profit business, a not-for-profit (NFP) social enterprize, or a hybrid model such as...
-
What tax is both withheld from an employee's pay and matched by the employer?
-
Identify the limitations that may be imposed in a variable interest entity. how these limitation be imposed?
-
My team - Director Nominated supervisor Educational leader Two room leaders 1 educator 4 trainee's Question Describe the structure of the team in your service. (e.g. Director/manager, administrative...
-
You have 505mL of a 0.125 M HCl solution and you want to dilute it to exactly 0.100 M. How much water should you add? Assume volumes are additive.
-
On 1 July 2021, Croydon Ltd leased ten excavators for five years from Machines4U Ltd. The excavators are expected to have an economic life of 6 years, after which time they will have an expected...
-
What aromatic products would you obtain from the KMnO4 oxidation of the followingsubstances? (a) O2N. (b) C(CH3)3 CH(CH3)2 "
-
Refer to Table 5.3 for quantitative idea of the stability of a benzyl radical. How much more stable (in kJ/mol) is the benzyl radical than a primary alkyl radical? How does a benzyl radical compare...
-
Styrene, the simplest alkenylbenzene, is prepared commercially for use in plastics manufacture by catalytic dehydrogenation of ethylbenzene. How might you prepare styrene from benzene using reactions...
-
A scuba diver is exploring a shipwreck at a depth of 30 meters in the ocean. The diver's tank contains 5.0 moles of compressed air at a temperature of 25C. What is the pressure of the air in the tank...
-
1. Determine the atmospheric pressure at a location where the barometric reading is 720 mm Hg. 2. A pressure gauge connected to a tank reads 52 psi at a location where the barometric reading is 29.6...
-
Assume that a bond has an annual coupon rate of 6.80 percent, but makes coupon payments on a semi-annual basis, has a par value of $1,000, a yield to maturity of 7.20 percent, and a current price of...

Study smarter with the SolutionInn App


