Use the Gibbs phase rule to determine the number of degrees of freedom in each region of
Question:
Use the Gibbs phase rule to determine the number of degrees of freedom in each region of the phase diagram in Figure 11-6.
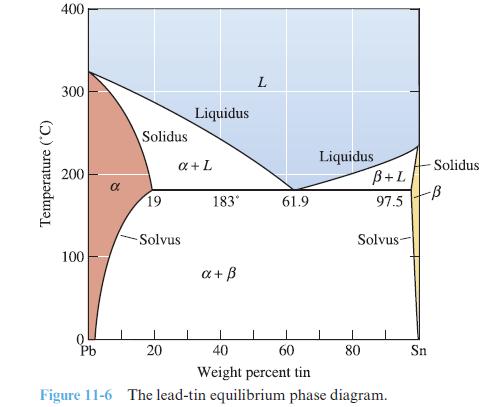
Transcribed Image Text:
Temperature (°C) 400 300 200 100 Pb Solidus 19 -Solvus 20 Liquidus a + L 183° a+ ß L 61.9 Liquidus B+L 97.5 Solvus 80 60 40 Weight percent tin Figure 11-6 The lead-tin equilibrium phase diagram. Solidus -ß Sn
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
The Gibbs phase rule is given by F C P 2 where F is the number ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Use the multiplicative rule to determine the number of sample points in the sample space corresponding to the experiment of tossing a coin the following number of times: a. 2 times b. 3 times c. 5...
-
Determine the number of degrees of freedom for a nonadiabatic equilibrium flash for one liquid feed, one vapor stream product, and two immiscible liquid stream products as shown inFigure. V.y, Vapor...
-
Determine the number of degrees of freedom for the two-sample t test or CI in each of the following situations: a. m = 10, n = 10, s1 = 5.0, s2 = 6.0 b. m = 10, n = 15, s1 = 5.0, s2 = 6.0 c. m = 10,...
-
Homzmart was founded in late 2019 by Daraz's former COO Mahmoud Ibrahim and Ibrahim Mohammed who led Jumia's h homzmart logistics development in Egypt to make furniture discovery and shopping easier...
-
(a) Redraw demand curve for pounds D£ and supply curve of pounds S£ as in the figure of Problem 1 and draw on it another supply curve for pounds (label it S*£) that intersects...
-
Schmaltz Cable Company's balance sheet reports the following assets under Property, Plant, and Equipment: Land, Buildings, Office Furniture, Communication Equipment, and Televideo Equipment. The...
-
What three elements must be included in the caption of a complaint?
-
Although the equity method is a generally accepted accounting principle (GAAP), recognition of equity income has been criticized. What theoretical problems can opponents of the equity method...
-
QUESTION 1 Customers arrive at an ATM at a rate of 62 per hour (assume that the arrival process can be described by a Poisson Distribution). The average time a customer spends at the machine...
-
On 1st September 2021, HA was waiting for a friend next to a bus stop in Yewsbridge. The road by the bus stop is narrow and there have been accidents on that road before where pedestrians on the...
-
Consider an Al-12% Mg alloy (Figure 11-29). During solidification, determine (a) The composition of the first solid to form; (b) The liquidus temperature, solidus temperature, solvus temperature, and...
-
We discussed the primary phase or primary constituent. Why would we be interested in the percentage of the primary phase in the Al-Si alloy system?
-
Jana Morgan is about to sign up for cellular telephone service. She is primarily interested in the safety aspect of the phone; she wants to have one available for emergencies. She does not want to...
-
One of the original aims of the Seventh Directive was to assist with the supervision of multinational enterprises by their host countries. Examine and discuss arguments for and against such a desire...
-
Two tuning forks are struck simultaneously, the first one having a frequency of \(762 \mathrm{~Hz}\). If a nearby listener hears 20 beats per second, determine the possible frequency or frequencies...
-
Two violin players are next to each other on a theater's stage. One plays a \(350-\mathrm{Hz}\) note, while the other simultaneously plays a \(353-\mathrm{Hz}\) note. (a) What is the beat frequency...
-
Two stationary tuning forks with frequencies of \(246 \mathrm{~Hz}\) and \(252 \mathrm{~Hz}\) are struck simultaneously. Calculate the frequency of the resulting sound. \(\cdot\)
-
Radio Station DXYZ has a power output of 128 W. While listening at home, you measure the intensity of the sound to be \(3.40 \times 10^{-6} \mathrm{~W} / \mathrm{m}^{2}\). How far away from your home...
-
PowerTap Utilities is planning to issue bonds with a face value of $1,000,000 and a coupon rate of 10 percent. The bonds mature in 10 years and pay interest semiannually every June 30 and December...
-
Express mass density in kg/m3 and weight density in lb/ft3. 1. Find the mass density of a chunk of rock of mass 215 g that displaces a volume of 75.0 cm3 of water. 2. A block of wood is 55.9 in. x...
-
Identify the Lewis acids in the following reactions: a. b. BF3 + NH3 FB-NH3
-
Gaseous SO 2 is created by combustion of sulfur-containing fuels, especially coal. Explain how O 2 in the atmosphere makes acidic rain.
-
Identify the Brnsted-Lowry acids among the reactants in the following reactions: a. b. KCN + HI = HCN + KI
-
Discuss and compare the strategies of a key competitor to Ulta Beauty. Discuss important differences and similarities in strategy. What does the competitor's future look like?
-
Coca-Cola introducing a new line of chai tea is an example of which market-product strategy?
-
What purpose do sweatshops serve in the fashion industry? 6. Here is a representation of the fashion apparel sold in Canada over the last 26+ years. In 1989, 70% of apparel sold in Canada was made...

Study smarter with the SolutionInn App


