Use the Lewis acidbase model to explain the following reaction. CO(g) + HO(1) HCO3(aq)
Question:
Use the Lewis acid–base model to explain the following reaction.
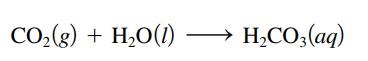
Transcribed Image Text:
CO₂(g) + H₂O(1) H₂CO3(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
The Lewis acidbase model explains chemical reactions in terms of the transfer of electrons from a Le...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Propose a mechanism to explain the following reaction by using words, and in doing so predict the absolute stereochemistry (R or S) at the stereocentre in the product. Then draw three other product...
-
Use Lewis symbols to show the reaction of atoms to form arsine, AsH3. Indicate which electron pairs in the Lewis formula of AsH3 are bonding and which are lone pairs.
-
The following are representations of acid base reactions: a. Label each of the species in both equations as an acid or a base and explain. b. For those species that are acids, which labels apply:...
-
At year-end 2010, 28,879 million represents: A. the funded status of the plan. B. the defined benefit obligation. C. the fair value of the plans assets. Kensington plc, a hypothetical company based...
-
Explain the four major principles of risk classification.
-
A sealed rigid vessel has volume of 1 m3 and contains 2 kg of water at 100C. The vessel is now heated. If a safety pressure valve is installed, at what pressure should the valve be set to have a...
-
Journalize the following transactions for Amazing Audio, Inc., that occurred during the month of November. Amazing Audios cost of inventory is 65% of the sales price. Nov 3 Sold $1,600 of merchandise...
-
On December 31, 2017, Dow Steel Corporation had 600,000 shares of common stock and 300,000 shares of 8%, noncumulative, nonconvertible preferred stock issued and outstanding. Dow issued a 4% common...
-
Damien Mellick and Hemi Nikora share a small apartment over a commercial shopfront in Albany, New Zealand, only a short walk from the post office. Hemi works part-time as an IT consultant for a local...
-
On January 1, 2022, Jarrison Mining Company purchased a piece of land for mining. The land cost $600,000, including all costs to ready the land for the operation. Jarrison expects 200,000 tons to be...
-
Is an aqueous solution of NaHSO 4 acidic, basic, or neutral? What reaction occurs with water? Calculate the pH of a 0.10-M solution of NaHSO 4 .
-
Calculate the pH of a 0.010-M solution of iodic acid (HIO 3 , K a = 0.17).
-
What is meant by the "institutionalization" of capital markets?
-
Determine the steady-state amplitudes for the equation \[\ddot{x}+0.05 \dot{x}+x+0.01 x^{3}=0.03 \sin 1.01 t\]
-
What is \(P(0)\) for the normalized Gaussian distribution?
-
A narrowband process has a power spectral density defined over a narrow band of frequencies. Indicate whether the statement presented is true or false. If true, state why. If false, rewrite the...
-
A beam element has four degrees of freedom. Indicate whether the statement presented is true or false. If true, state why. If false, rewrite the statement to make it true.
-
The boundary conditions at a free end for a beam are natural boundary conditions. Indicate whether the statement presented is true or false. If true, state why. If false, rewrite the statement to...
-
Describe what is included in shortage costs.
-
For the given transfer function: Vo(s) / Vi(s) = (s^2C^2R^2 + 1) / (s^2C^2R^2 + 4sCR + 1) Assumiing that 1/(CR) = 120 PI so write the matlab code to find the magnitude plot
-
The hydrolysis of a biological thioester to the corresponding carboxylate is often more complex than the overall result might suggest. The conversion of succinyl CoA to succinate in the citric add...
-
One step in the gluconeogenesis pathway for the biosynthesis of glucose is the partial reduction of 3-phosphoglycerate to give glyceraldehydes 3-phosphate. The process occurs by phosphorylation with...
-
Penicillins and other fl-lactam antibiotics (see the Focus On in this chapter) typically develop a resistance to bacteria due to bacterial synthesis of -lactamase enzymes. Tazobactam, however, is...
-
At the end of the current year, Accounts Receivable has a balance of $880,000, Allowance for Doubtful Accounts has a credit balance of $8,000, and sales for the year total $3,960,000. Bad debt...
-
On January 1 of this year, Ikuta Company issued a bond with a face value of $120,000 and a coupon rate of 5 percent. The bond matures in 3 years and pays interest every December 31. When the bond was...
-
Discuss the impact of enzyme engineering and directed evolution in the development of enzymes with enhanced or novel catalytic activities for industrial or pharmaceutical applications .

Study smarter with the SolutionInn App


