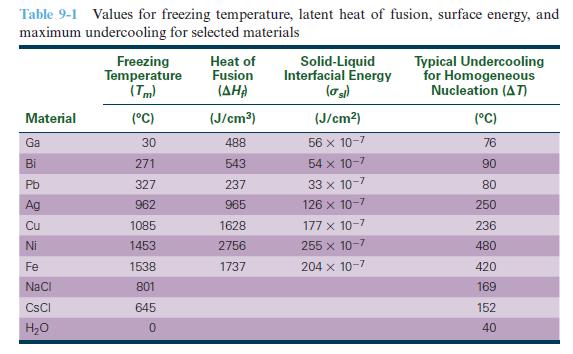
Using the densities in Appendix A, convert the heats of fusion in Table 9-1 from units of
Question:
Using the densities in Appendix A, convert the heats of fusion in Table 9-1 from units of J/cm3 to kJ/kg.
Transcribed Image Text:
Table 9-1 Values for freezing temperature, latent heat of fusion, surface energy, and maximum undercooling for selected materials Material Ga Bi Pb Ag 23 22 Cu Ni Fe NaCl CsCl H₂O Freezing Temperature (Tm) (°C) 30 271 327 962 1085 1453 1538 801 645 0 Heat of Fusion (AHA) (J/cm³) 488 543 237 965 1628 2756 1737 Solid-Liquid Interfacial Energy (σ sl) (J/cm²) 56 x 10-7 54 x 10-7 33 x 10-7 126 x 10-7 177 x 10-7 255 x 10-7 204 x 10-7 Typical Undercooling for Homogeneous Nucleation (AT) (°C) 76 90 80 250 236 480 420 169 152 40
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
QUESTION 1 Aluminum has a density of 1.35 g/cm. What is the mass of a rectangular block of aluminum measuring 11.1 cm by 22.2 cm by 34.6 cm? O a. none of these Ob.0.159 kg O c. 11.5 kg O d. 183 kg...
-
Calculate the acceleration of the center of mass of the system of the four 10-kg cylinders. Neglect friction and the mass of the pulleys and cables. 500 N 250 N 10 10 kg kg 10 kg 10 kg
-
Using the heats of fusion and vaporization for water, calculate the change in enthalpy for the sublimation of water: H2O(s) H2O(g) Using the H value given in Exercise 24 and the number of hydrogen...
-
You are considering an investment that will be valued at $1,000 one year from now. Your next best alternative investment opportunity would yield an annual rate of return of 8%, In other words, if you...
-
In April 2000, the seasonally adjusted unemployment rate was 3.8 percent. By June 2001, the unemployment rate had increased to 4.5 percent. Yet the measures by the Federal Reserve to reduce...
-
The students receiving bachelor of science degrees from a university include 52 biology majors. In Exercises 58, determine whether the number describes a population parameter or a sample statistic....
-
Compute the following items for the statement of cash flows: a. Beginning and ending Retained Earnings are \($45,000\) and \($70,000,\) respectively. Net income for the period is \($60,000.\) How...
-
Lloyd Industries manufactures electrical equipment from specifications received from customers. Job X10 was for 1,000 motors to be used in a specially designed electrical complex. The following costs...
-
The role of capital markets is paramount in the global economy, serving as the nexus where entities raise funds by issuing securities and investors allocate capital to various investment...
-
Cleary Foods produces speciality soup sold in jars. The projected sales in dollars and jars for each quarter of the upcoming year are as follows: Total sales revenue 1st quarter....$ 181,000 2nd...
-
Explain the meaning of each term in Equation 9-2. 20. Tm , (9-2)
-
What is the difference between homogenous nucleation and heterogeneous nucleation?
-
How will the Santa Fe Grill owners be able to use the social media usage patterns of the respondents to develop a more effective marketing plan for their restaurant?
-
You own 350 shares of Voestalpine AGs preferred stock at a market price of \($32\) per share. Voestalpine AG pays a dividend of \($1.65.\) What is your expected rate of return? If you have a required...
-
You own 250 shares Magna Steyr AGs preferred stock, which currently sells for \($55\) per share and pays an annual dividend of \($4.20\) per share. a. What is your expected return? b. If you require...
-
Bugattis return on equity is 15 percent, and the management plans to retain 50 percent of earnings for investment purposes. What will be the firms growth rate?
-
Define the term cost of capital.
-
Bestattung Wiens preferred stock is selling for \($43.29\) per share and pays \($2.10\) in dividends. What is your expected rate of return if you purchase the security at market price?
-
The average return for large-cap domestic stock funds over the three years 2009 - 2011 was 14.4% (AAII Journal, February, 2012). Assume the three-year returns were normally distributed across funds...
-
-4 1 9. Let A = Find A-1, (A") and verify that (A")= (A-1)".
-
Which of the following mixtures would result in a buffered solution when 1.0 L of each of the two solutions are mixed? a. 0.2 M HNO 3 and 0.4 M NaNO 3 b. 0.2 M HNO 3 and 0.4 M HF c. 0.2 M HNO 3 and...
-
Which of the following mixtures would result in buffered solutions when 1.0 L of each of the two solutions are mixed? a. 0.1 M KOH and 0.1 M CH 3 NH 3 Cl b. 0.1 M KOH and 0.2 M CH 3 NH 2 c. 0.2 M KOH...
-
Calculate the pH of a solution that is 0.20 M HOCl and 0.90 M KOCl. In order for this buffer to have pH = pK a , would you add HCl or NaOH? What quantity (moles) of which reagent would you add to 1.0...
-
Consider a uniformly charged ring of radius 1.2 m and total charge Q = -3 C placed on the origin of the x axis as shown in the picture below. a. Determine magnitude and direction of the electric...
-
Allan Bakke, a thirty-five-year-old white man, had twice applied for admission to the University of California Medical School at Davis. He was rejected both times. The school reserved sixteen places...
-
Healthcare managers are involved in the annual budget and must have a good understanding of the financial statements and how they impact their individual departments. Capital budget proposals are...

Study smarter with the SolutionInn App


