We expect the atomic radius to increase going down a group in the periodic table. Can you
Question:
We expect the atomic radius to increase going down a group in the periodic table. Can you suggest why the atomic radius of hafnium breaks this rule? (See data below.)
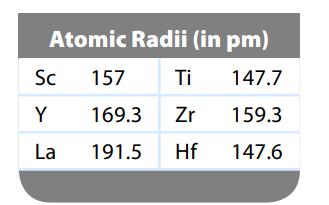
Transcribed Image Text:
Atomic Radii (in pm) 147.7 159.3 147.6 Sc Y La 157 169.3 191.5 Ti Zr Hf
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 46% (13 reviews)

Answered By

Shebla K
I am an MBA graduate having experience as an Assistant Professor at University level for two years. I always prepare well for a class as I believe that only if you become an ocean you can give a bucket of water. Being a teacher was not only my profession but also my passion.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
To what group in the periodic table would an element with atomic number 114 belong?
-
To what group in the periodic table would an element with atomic number 114 belong?
-
The changes in electron affinity as one goes down a group in the periodic table are not nearly as large as the variations in ionization energies. Why?
-
Featherstone Inc. reported the following data: Net income ................................... $296,000 Depreciation expense ................... 113,100 Gain on disposal of equipment ...... 58,500...
-
Valencia Products makes automobile radar detectors and assembles two models: LaserStop and SpeedBuster. The firm can sell all it produces. Both models use the same electronic components. Two of these...
-
About what percent of x values lie between the first and second standard deviations from the mean (both sides)?
-
From the following accounts of Resch Restore, Inc., prepare the businesss statement of retained earnings for the year ended January 31, 2010: Retained Earnings Dividends Clo Clo 110,000 Feb 1 82,000...
-
What are the two elements that an employee must show to make a successful Title VII claim? Do you think the motivating factor standard is appropriate for discrimination claims based on national...
-
Student enrollment is up at the local community college. Lacey Grey, the admissions and enrollment coordinator is excited about this, but she is also worried that the current enrollment process may...
-
? ? Please complete the 2019 federal income tax return for Joseph and Diana Cohen. Ignore the requirement to attach the form(s) W-2 to the front page of the Form 1040. If required information is...
-
Calculate, to four significant figures, the longest and shortest wavelengths of light emitted by electrons in the hydrogen atom that begin in the n = 5 state and then fall to states with smaller...
-
The four most abundant elements by mass in the human body are oxygen, carbon, hydrogen, and nitrogen. These four elements make up about 96% of the human body. The next four most abundant elements are...
-
Would there ever be activities that relate to operating, investing, or financing activities that would not be reported in their respective sections of the statement of cash flows? Explain. If a...
-
How would one determine the extent to which locals are reverse time-switchers for a particular event?
-
Based on the answers to questions 15, should the project be accepted? Why or why not?
-
All else being equal, does increasing the size of the geographic area of impact raise, lower, or have no effect on the capture rate?
-
What is the projects NPV?
-
Under what conditions should spending by local residents be counted in a calculation of economic impact?
-
Accounting records for Ontario Corporation yield the following data for the year ended June 30, 2016 (assume sales returns are non-existent): Inventory, June 30, 2015...
-
A company produces earbuds. The revenue from the sale of x units of these earbuds is R = 8x. The cost to produce x units of earbuds is C = 3x + 1500. In what interval will the company at least break...
-
The systematic name of the ? CH = CH2 group is ethenyl: Provide a systematic name for limonene, which is found in lemons and other citrus-fruits. Limonene
-
Vitamin A alcohol is 3, 7 dimethyl-9-(2, 6, 6-trimethyl-1-cyclohexenyl)-2, 4, 6, 8-nonatetraen-1-o1, Draw the stricter of vitamin A alcohol.
-
Explain which compound has the higher melting point: (a) Cyclopentane or pentane (b) 1-Pentanol or pentane
-
Explained the different type of leader -Great man -trait theory -skill theory -behavior theory -Contingency theory (situational) -Transformation transactional leadership -transactional leadership...
-
What effect will a corporation's emphasis on environmental responsibility have on its financial performance in both the short term and long term? Does this contradict the notation of maximizing the...
-
Examine the emergence of technology and electronic health systems in health care since the passage of the Health Insurance Portability and Accountability Act (HIPAA). Provide an analysis of the...

Study smarter with the SolutionInn App


