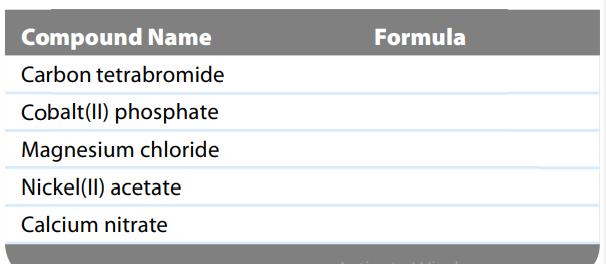
What are the formulas of the compounds that correspond to the names given in the following table?
Question:
What are the formulas of the compounds that correspond to the names given in the following table?
Transcribed Image Text:
Compound Name Carbon tetrabromide Cobalt(II) phosphate Magnesium chloride Nickel(II) acetate Calcium nitrate Formula
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
1 C Br 4 2 Co 3 ...View the full answer

Answered By

DHRUV RAI
As a tutor, I have a strong hands-on experience in providing individualized instruction and support to students of all ages and ability levels. I have worked with students in both one-on-one and group settings, and I am skilled in creating engaging and effective lesson plans that meet the unique needs of each student.
I am proficient in using a variety of teaching techniques and approaches, including problem-based learning, inquiry-based learning, and project-based learning. I also have experience in using technology, such as online learning platforms and educational software, to enhance the learning experience for my students.
In addition to my teaching experience, I have also completed advanced coursework in the subjects that I tutor, including mathematics, science, and language arts. This has allowed me to stay up-to-date on the latest educational trends and best practices, and to provide my students with the most current and effective teaching methods.
Overall, my hands-on experience and proficiency as a tutor have equipped me with the knowledge, skills, and expertise to help students achieve their academic goals and succeed in their studies.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
What are the names of the compounds that correspond to the formulas given in the following table? Formula Co(NO)2 AsF5 LICN KSO3 Li3N PbCrO4 Compound Name
-
Explain the Component Diagram of Bug Tracker in detail.
-
The table below gives the break and halt temperatures found in the cooling curves of two metals A and B. Construct a phase diagram consistent with the data of these curves. Label the regions of the...
-
A social skills training program was implemented with seven mildly challenged students in a study to determine whether the program caused improvements in pre/post measures and behavior ratings. For...
-
Rachel Schurtz is a high school English teacher. In her spare time, she likes to make her own body lotion, lip-gloss, and bath and shower gel. She uses the bath products herself and gives them to her...
-
Average daily insolation in Phoenix is 5.38 kWh/m2/day. Given this daily energy input, how much land area would you need (in square miles) to generate all of Arizona's daily electricity from the...
-
The equation of motion of a single-degree-of-freedom system is given by \[2 \ddot{x}+0.8 \dot{x}+1.6 x=0\] with initial conditions \(x(0)=-1\) and \(\dot{x}(0)=2\). (a) Plot the graph \(x(t)\) versus...
-
Under U. S. GAAP, property, plant, and equipment are reported at historical cost net of accumulated depreciation. These assets are written down to fair value when it is determined that they have been...
-
Thomas Cycles manufactures chainless bicycles. On March 31, Thomas Cycles had 214 bikes in inventory. The company has a policy that the ending inventory in any month must be 20% of the following...
-
Aggressive versus Conservative seasonal funding strategy Dynabase Tool has forecast its total funds requirement for the coming year as shown in the following table. a. Divide the firm's monthly funds...
-
By analogy with phosphorus compounds, name the following: Na 3 AsO 4 , H 3 AsO 4 , Mg 3 (SbO 4 ) 2 .
-
An elements most stable ion forms an ionic compound with bromine, having the formula XBr 2 . If the ion of element X has a mass number of 230 and has 86 electrons, what is the identity of the...
-
For the rigid frame shown in Figure P5-4, determine (1) the nodal displacements and rotation at node 4, (2) the reactions, and (3) the forces in each element. Then check equilibrium at node 4....
-
It is March 2022, and you receive notice from your customer that there will be around April 2023 for an advanced component to be integrated into the current system acquired by your agency last year....
-
Help getting only city of each country by using this indices, City_indices = [0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11] cities = [{'City': 'Buenos Aires', 'Country': 'Argentina', 'Population': 2891000,...
-
Part 1: Concise output In this section, we will modify the program in Lab 1 to produce more concise output. In CSV (comma-separated values) format, values are separated by commas (hence the name)....
-
What physical changes occur, in general, as a person ages? How can this affect their work? Why should a workplace look at issues concerning aging workers? How does the way older people learn and...
-
Use theoretical and economic analysis to examine public policy or administration issues, considering political economy and economic factors. Analysis of decision-making, management, organizational,...
-
On January 4, 2016, Spandella Company purchased 175,000 shares of Filington Company directly from one of the founders for a price of $30 per share. Filington has 500,000 shares outstanding, including...
-
A 20-cm-square vertical plate is heated to a temperature of 30oC and submerged in glycerin at 10oC. Calculate the heat lost from both sides of the plate.
-
Explain how the mixing of reactants and products affects the position of chemical equilibrium.
-
Account for Le Chatelier's principle in terms of thermodynamic quantities.
-
(a) How may an Ellingham diagram be used to decide whether one metal may be used to reduce the oxide of another metal? (b) Use the Ellingham
-
Athens Gas Station has figured out the weekly demand distribution for their gas sales. Each gallon of gas sold at the pump results in a profit of 10 cents/gallon and any lost sales results in a cost...
-
1. The maximum pressure an eardrum can withstand without rupturing is Pmax = 3.0102 Pa. Assume a sound is made at that pressure. If the density of air is, p=1.20 kg/m, and the speed of sound is 340...
-
52. A wave is sent down a string with a linear mass density of 1.600 x 10-4 kg/m. The wavefunction that describes this wave is: y(x,t) = (0.021 m)*sin{(2.00 rad/m)*x + (30.0 rad/s)*t} What is the...

Study smarter with the SolutionInn App


