What type of molecular orbital would result from the in-phase combination of two d xz atomic orbitals
Question:
What type of molecular orbital would result from the in-phase combination of two dxz atomic orbitals shown below? Assume the x-axis is the internuclear axis.
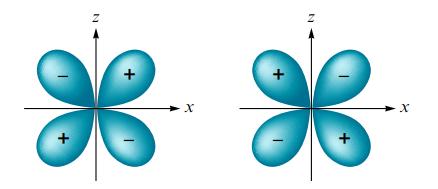
Transcribed Image Text:
z Z X X X
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
When the two electrons come together and they are out of ...View the full answer

Answered By

Hardik Dudhat
I am semi-qualified Chemical Engineering ,I have scored centum in accounting in my senior secondary and in my graduation. I have always helped my fellow students with their concerns on the subject, i have tutored on various tutoring sites in the past and also have taken home tuitions for degree and MBA students. As a tutor, I don't want my students to just get a solution, I want them to understand the concept and never have a doubt in that area thereon and i believe in excelling and not in educating.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
The following sketches show the atomic orbital wave functions (with phases) used to construct some of the MOs of a homo-nuclear diatomic molecule. For each sketch, determine the type of MO that will...
-
The sp2 hybrid atomic orbitals have the following general form: where Ïs, Ïpx, and Ïpy represent orthonormal (normalized and orthogonalized) atomic orbitals. Calculate the values of A...
-
Assume that the cyanide ion, CN, has molecular orbitals similar to those of a homonuclear diatomic molecule. Write the configuration and bond order of CN. Is a substance of the ion diamagnetic or...
-
Roles define the manager. Do you agree or disagree with this statement? Discuss what you think managers do.
-
Why do firms divest assets?
-
Write structural formulas for the cyclohexadienyl cations formed from aniline (C6H5NH2) during (a) Ortho bromination (four resonance structures) (b) Meta bromination (three resonance structures) (c)...
-
Attracted by the possible returns from a portfolio of movies, hedge funds have invested in the movie industry by financially backing individual films and/or studios. The hedge fund Star Ventures is...
-
You have just completed the first year of operation for your business and have the following information: sales, $200,000; cost of goods, $140,000; rent, $18,000; utilities, $8,400; insurance,...
-
Describe the situation from either your professional experience or your research in which unethical or fraudulent behavior occurred. If you are describing an example from experience, please do not...
-
Russian is an Indo-European language of the Slavic family, spoken in Russia. Determine from the following Russian data whether the low front [a] and the low back [a] complement each other as...
-
Using molecular orbital theory, explain why the removal of an electron from O 2 strengthens bonding, whereas the removal of an electron from N 2 weakens bonding.
-
The transport of O 2 in the blood is carried out by hemoglobin. Carbon monoxide (CO) can interfere with O 2 transport because hemoglobin has a stronger affinity for CO than for O 2 . If CO is...
-
According to Keynes, an increase in saving and decrease in consumption may lower total spending in the economy. But how could this happen if the increased saving lowers interest rates (as shown in...
-
The risk-free rate of return is 4% and the expected return on the market is 9.2%. Stock A has a beta coefficient of 1.5 and dividend growth rate 5% Current dividend is $2.10 a share. What would be...
-
Write a paragraph about the role of color psychology in marketing and business.Write a paragraph about the role of color psychology in marketing and business.
-
Based on the Lease Asset vs Total Asset visualization, what conclusions can be made?
-
How does Positive Psychology differ from Clinical Psychology? Please add references.
-
1) Discuss why you feel it is important for a business owner to understand the connections between the financial statements. 2) Give an example of how regular analysis of the information might assist...
-
Hydrogen fluoride is used in the manufacture of Freons (which destroy ozone in the stratosphere) and in the production of aluminum metal. It is prepared by the reaction In one process, 6.00 kg of...
-
Estimate a range for the optimal objective value for the following LPs: (a) Minimize z = 5x1 + 2x2 Subject to X1 - x2 3 2x1 + 3x2 5 X1, x2 0 (b) Maximize z = x1 + 5x2 + 3x3 Subject to X1 + 2x2 +...
-
What is the difference between a transition state and an intermediate?
-
Draw an energy diagram for a one-step reaction with Keq < 1. Label the parts of the diagram corresponding to reactants, products, transition state, G, and G++. Is G positive or negative?
-
Draw an energy diagram for a two-step reaction with Keq > 1. Label the overall G, transition states, and intermediate. Is G positive or negative?
-
A 2-meter-tall basketball player attempts a goal 12.4 meters from the basket that is 3.05 meters high. If he shoots the ball at a 55 angle, at what initial speed must he throw the basketball so that...
-
Find the following. 2. 3 y(t) + 3y(t) 3+ = x(t) + 3 x(t) 3. y(t) - 1/2 y(t) x(t) = dt d 4. dt d 5. dt y(t) + 1/2 y(t) + y(t) y(t) + 1/2 y(t) = x(t) + x(t) = x(t)
-
1. Who are the people affected by rsum truth decisions? 2. Why might a job seeker have a duty to blur parts of his or her work history? 3. Why might an egoist lie on the rsum, and why not? 4. Does an...

Study smarter with the SolutionInn App


