Calculate the HHV and LHV of gaseous n-octane fuel (C 8 H 18 ). Compare your results
Question:
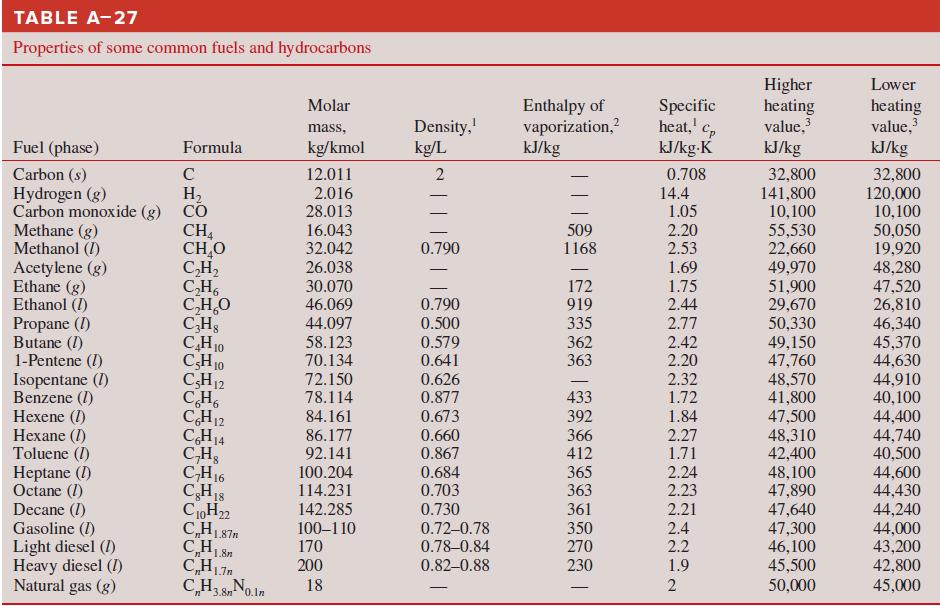
Calculate the HHV and LHV of gaseous n-octane fuel (C8H18). Compare your results with the values in Table A–27.
Transcribed Image Text:
TABLE A-27 Properties of some common fuels and hydrocarbons Higher heating value, Lower Enthalpy of vaporization,? kJ/kg Specific heat,' kJ/kg-K Molar Density, kg/L heating value, kJ/kg 2 mass, Fuel (phase) Formula kg/kmol kJ/kg Carbon (s) Hydrogen (g) Carbon monoxide (g) CO Methane (g) Methanol (I) Acetylene (g) Ethane (g) Ethanol (I) Propane (I) Butane (I) 1-Pentene (I) Isopentane (1) Benzene (1) Hexene (1) Hexane (I) Toluene (I) Heptane (I) Octane (1) Decane (I) Gasoline (I) Light diesel (I) Heavy diesel (I) Natural gas (g) 32,800 141,800 10,100 55,530 22,660 49,970 51,900 29,670 50,330 49,150 47,760 48,570 41,800 C 12.011 0.708 32,800 120,000 10,100 50,050 19,920 48,280 47,520 26,810 46,340 45,370 44,630 44,910 40,100 H, 2.016 28.013 14.4 1.05 CH, CH,O C,H, C,H, C,H,O CHs CH10 CH10 CH12 CH, CH12 CH14 C,H, CH16 CH18 C0H22 C,H187m 16.043 32.042 26.038 509 1168 2.20 0.790 2.53 1.69 30.070 46.069 172 919 1.75 0.790 2.44 335 362 363 44.097 0.500 2.77 58.123 70.134 72.150 0.579 0.641 2.42 2.20 2.32 0.626 0.877 433 392 366 412 78.114 1.72 84.161 0.673 1.84 47,500 48,310 42,400 48,100 47,890 47,640 47,300 46,100 45,500 44,400 44,740 40,500 44,600 44,430 44,240 44,000 43,200 42,800 86.177 0.660 2.27 1.71 2.24 92.141 0.867 100.204 114.231 0.684 0.703 365 363 2.23 142.285 0.730 361 2.21 100–110 170 0.72-0.78 0.78-0.84 350 270 2.4 2.2 200 0.82–0.88 230 1.9 CH3 8No.ln C,H3,8n0.ln 50,000 45,000 18
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The higher and lower heating values of gaseous octane are to be determined and compared to the liste...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Engineering questions
-
Calculate the HHV and LHV of liquid propane fuel (C3H8). Compare your results with the values in Table A-27 109258336914756 4202 367073567 88156996317585341863150 21052919097817828777650...
-
Calculate the higher and lower heating values of gaseous methane fuel (CH 4 ). Compare your results with the values in Table A27.
-
Compare your results with those obtained in Problem 8.113 (p. 330)? Microbiology Refer to the data in Table 8.28 (p. 330). Table 8.28 Pod weight (g) from inoculated (I) and uninoculated (U) plantsa
-
Write the nodal equations for the networks in Figure. Using determinants, solve for the nodal vo ltages. R4 2 5 A R 1 R3 50 4 3 A
-
A student experiences difficulties with malfunctioning alarm clocks. Instead of using one alarm clock, he decides to use three. What is the probability that at least one alarm clock works correctly...
-
Selected accounts for Lee?s Salon at December 31, 2021, are presented below. Instructions a. Prepare closing entries to close the accounts. b. Post the entries. A. Lee, Capital A. Lee, Drawings Cr....
-
A normal shock propagates at \(2000 \mathrm{ft} / \mathrm{s}\) into the still air in a tube. The temperature and pressure of the air are \(80^{\circ} \mathrm{F}\) and 14.7 psia before "hit" by the...
-
The following financial data were reported by 3M Company for 2009 and 2010 (dollars in millions). Instructions (a) Calculate the current ratio and working capital for 3M for 2009 and 2010. (b)...
-
A small mining group is considering the purchase of a $1,786,713 continuous mining machine that will operate approximately 6,000 hours per year. The operational costs for the unit are estimated to be...
-
Write a class that implements a Square shape. Class Square should contain a Side property for accessing. Private data Provide a constructor that takes a side length as a value. Also Provide the...
-
Repeat Prob. 1546 for liquid octane (C 8 H 18 ). Data From Q#46: Determine the enthalpy of combustion of methane (CH 4 ) at 25C and 1 atm, using the enthalpy of formation data from Table A26. Assume...
-
Propane fuel (C 3 H 8 ) is burned in a space heater with 50 percent excess air. The fuel and air enter this heater steadily at 1 atm and 17C, while the combustion products leave at 1 atm and 97C....
-
Perform the indicated operations, expressing all answers in the form a + bj. j2 - 5 j2 + 3
-
True Or False In deciding whether the tort of interference with existing contractual relations has been committed, courts consider the purpose and motive of the defendant as well as the social...
-
What have some states done in an effort to reduce the number of medical malpractice claims?
-
Why is nuisance referred to as the legal garbage can?
-
Under what circumstances is a plaintiff justified in relying on a defendants opinion?
-
True Or False Recovery for public nuisance is allowed even if only the plaintiff is injured, as long as that injury occurs in a public place.
-
An excerpt from Section 10.2 of the Management Discussion and Analysis in the 2014 annual report of BCE Inc. is shown below. The excerpt shows summarized financial information, including calculations...
-
Banner Company acquires an 80% interest in Roller Company for $640,000 cash on January 1, 2013. The NCI has a fair value of $160,000. Any excess of cost over book value is attributed to goodwill. To...
-
Determine the highest possible temperature that can be obtained when liquid gasoline (assumed C8H18) at 25oC is burned steadily with air at 25oC and 1 atm. What would your answer be if pure oxygen at...
-
Liquid propane (C3H8()) enters a combustion chamber at 25oC and 1 atm at a rate of 0.4 kg/min where it is mixed and burned with 150 percent excess air that enters the combustion chamber at 25oC. The...
-
Determine the work potential of 1 lbmol of diesel fuel (C12H26) at 77oF and 1 atm in an environment at the same state.
-
A project that costs $4,450 to install will provide annual cash flows of $1,500 for each of the next 6 years. a. What is NPV if the discount rate is 11%? b. How high can the discount rate be before...
-
Prove MM1: Both Umbrella Inc. and Prixxa Inc. produce $300,000 of cash flow each year. Umbrella has no debt outstanding, and its cost of equity capital is 14 percent. But Prixxa has 1,000,000 depts...
-
How did they find the over head rate percentages for current year and last year? A job order cost sheet for Cullumber Company is shown below. Job No. 92 For 2,000 Units Date Direct Materials Direct...

Study smarter with the SolutionInn App


