Determine the temperature at which 10 percent of diatomic hydrogen (H 2 ) dissociates into monatomic hydrogen
Question:
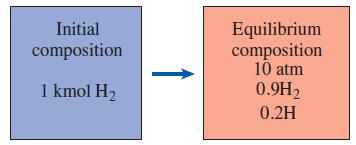
Determine the temperature at which 10 percent of diatomic hydrogen (H2) dissociates into monatomic hydrogen (H) at a pressure of 10 atm.
Transcribed Image Text:
Initial Equilibrium composition 10 atm 0.9H2 composition 1 kmol H2 0.2H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 45% (11 reviews)
The temperature at which 10 percent of H 2 dissociates into 2H is to be determined Assumptions 1 The ...View the full answer

Answered By

Kalyan M. Ranwa
I have more than seven years of teaching experience in physics and mechanical engineering.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:
Students also viewed these Engineering questions
-
Determine the temperature at which 5 percent of diatomic oxygen (O2) dissociates into monatomic oxygen (O) at a pressure of 3 atm.
-
Determine the temperature at which 5 percent of diatomic oxygen (O2) dissociates into monatomic oxygen (O) at a pressure of 3 atm.
-
A mixture of 1 mol of H2 and 1 mol of Ar is heated at a constant pressure of 1 atm until 15 percent of H2 dissociates into monatomic hydrogen (H). Determine the final temperature of the mixture.
-
101, 115, 143, 106, 100, 142, 157, 163, 155, 141, 145, 153, 152, 147, 143, 115, 164, 160, 147, 150 (90%) Find the confidence interval of the median, indicated in parentheses, for the set of data.
-
The amount of milk obtained from a cow
-
The unadjusted trial balance and adjustment data for Silver Ridge Plumbing are provided. Additional information: 1. The equipment has an expected useful life of 10 years. The vehicles? expected...
-
An expression for the value of \(c_{p}\) for carbon dioxide as a function of temperature is \[ c_{p}=286-\frac{1.15 \times 10^{5}}{T}+\frac{2.49 \times 10^{6}}{T^{2}} \] where \(c_{p}\) is in...
-
Helner Cell Phones (HCP) is developing a new touch screen smartphone to compete in the cellular phone industry. The phones will be sold at wholesale prices to cell phone companies, which will in turn...
-
Discuss some of the myths associated with teen pregnancy? Why are these myths incorrect and what harm can they do? How can we as workers in the fieldwork to help change these perceptions?
-
A stepped shaft ABC consisting of two solid circular segments is subjected to torques T1 and T2 acting in opposite directions, as shown in the figure. The larger segment of the shaft has diameter d1...
-
The equilibrium constant for C + 1/2 O 2 CO 2 reaction at 100 kPa and 1600 K is K p . Use this information to find the equilibrium constant for the following reactions at 1600 K. (a) C + 1/2 O 2 CO...
-
At what temperature will oxygen be 15 percent disassociated at (a) 3 psia (b) 100 psia?
-
Here is a forecast of sales by National Bromide for the first four months of 2010 (figures in $ thousands): On the average 50% of credit sales are paid for in the current month, 30% are paid in the...
-
Why might it be to a defendants advantage to discount an award of future damages?
-
True Or False Assumption of risk applies if plaintiff should have known of the risk.
-
What are the primary reasons for most legal malpractice claims?
-
What are differences between express and implied assumption of risk?
-
When sovereign immunity is abolished, a. it is done because courts believe being responsible for the torts of employees is one of the costs of administering a government. b. it is done even though...
-
Assume that Elrond Inc. decided to sell DemandTV Ltd., a subsidiary, on September 30, 2017. There is a formal plan to dispose of the business component, and the sale qualifies for discontinued...
-
1) The government decided to reduce taxes on fast-food to increase revenue. The government assumes that fast-food products have a) An inelastic demand b) An elastic demand c) A demand curve that is...
-
A constant-volume tank contains a mixture of 120 g of methane (CH4) gas and 600 g of O2 at 25oC and 200 kPa. The contents of the tank are now ignited, and the methane gas burns completely. If the...
-
Reconsider Prob. 15-65. Using EES (or other) software, investigate the effect of the final temperature on the final pressure and the heat transfer for the combustion process. Let the final...
-
One lbmol of methane (CH4) undergoes complete combustion with stoichiometric amount of air in a rigid container. Initially, the air and methane are at 14.4 psia and 77oF. The products of combustion...
-
What extent does emotional intelligence among leaders and employees contribute to effective teamwork and conflict resolution, and what methods can be employed to enhance emotional intelligence within...
-
Determine the exponential function y = c(b) that goes through the points (-2, 16) and (1,54). Algebraic work must be shown for full credit.
-
Solve 6. 8. 8 So log x (1 + x)(4+ x) [log x] 2 dx 0 1 + x So dx

Study smarter with the SolutionInn App


