Estimate the c p of nitrogen at 300 kPa and 400 K, using (a) The relation in
Question:
Estimate the cp of nitrogen at 300 kPa and 400 K, using
(a) The relation in Prob. 12–91,
Data From Q#91:
Consider an infinitesimal reversible adiabatic compression or expansion process. By taking s = s(P, v) and using the Maxwell relations, show that for this process Pvk = constant, where k is the isentropic expansion exponent defined as
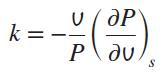
Also, show that the isentropic expansion exponent k reduces to the specific heat ratio cp/cv for an ideal gas.
(b) Its definition. Compare your results to the value listed in Table A–2b.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 9781259822674
9th Edition
Authors: Yunus Cengel, Michael Boles, Mehmet Kanoglu
Question Posted:





